Occurrence of Morpholine in Central Nervous System Drug Discovery
- PMID: 33459557
- PMCID: PMC7877733
- DOI: 10.1021/acschemneuro.0c00729
Occurrence of Morpholine in Central Nervous System Drug Discovery
Abstract
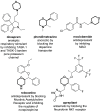
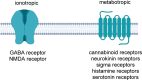
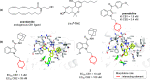
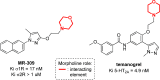
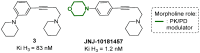
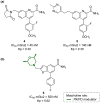
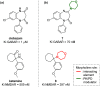
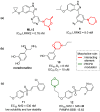
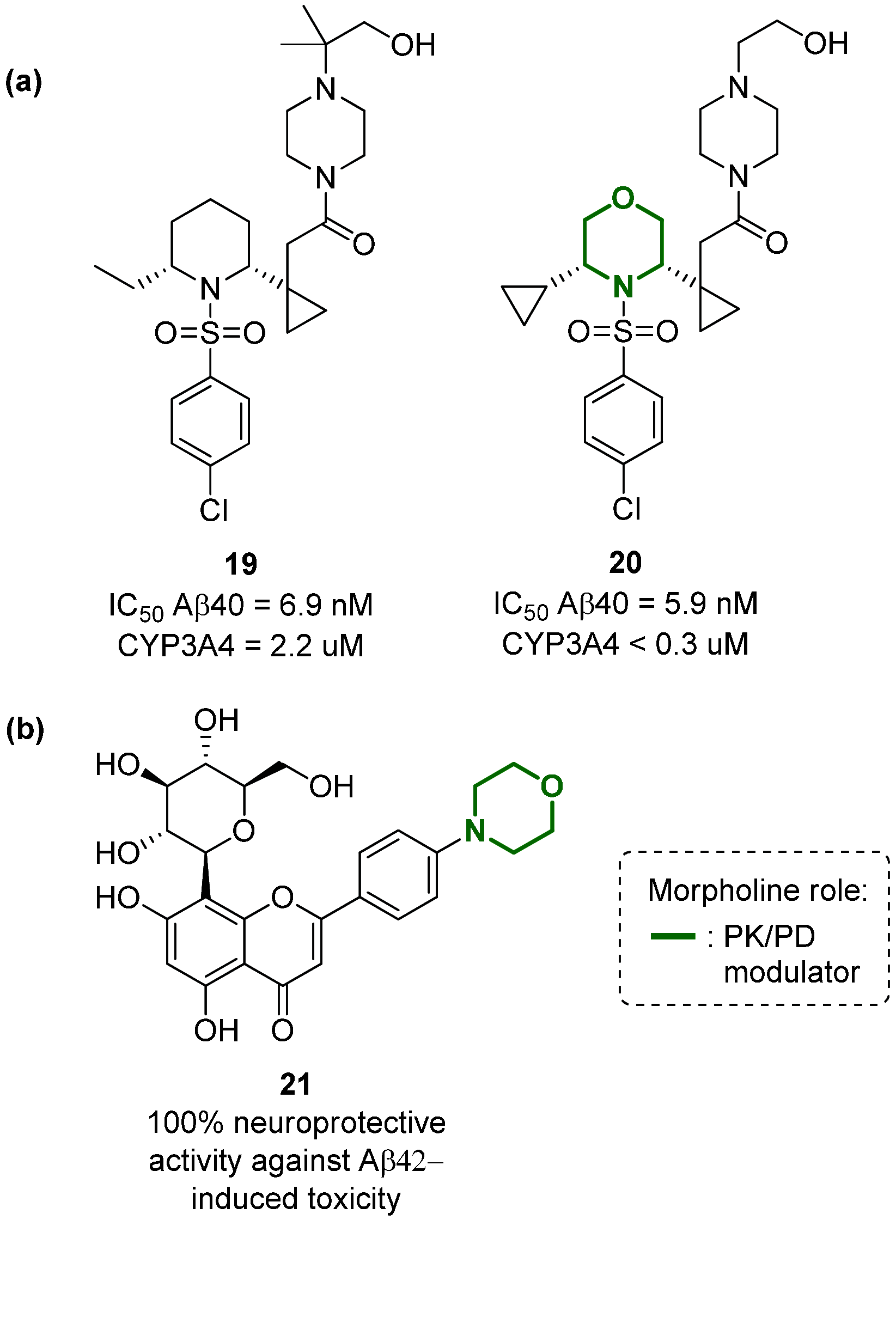
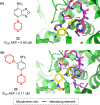
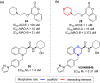
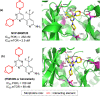
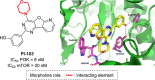
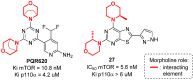
Developing drugs for the central nervous system (CNS) requires fine chemical modifications, as a strict balance between size and lipophilicity is necessary to improve the permeability through the blood-brain barrier (BBB). In this context, morpholine and its analogues represent valuable heterocycles, due to their conformational and physicochemical properties. In fact, the presence of a weak basic nitrogen atom and of an oxygen atom at the opposite position provides a peculiar pKa value and a flexible conformation to the ring, thus allowing it to take part in several lipophilic-hydrophilic interactions, and to improve blood solubility and brain permeability of the overall structure. In CNS-active compounds, morpholines are used (1) to enhance the potency through molecular interactions, (2) to act as a scaffold directing the appendages in the correct position, and (3) to modulate pharmacokinetic/pharmacodynamic (PK/PD) properties. In this perspective, selected morpholine-containing CNS drug candidates are discussed to reveal the active pharmacophores accountable for the (1) modulation of receptors involved in mood disorders and pain, (2) bioactivity toward enzymes and receptors responsible for neurodegenerative diseases, and (3) inhibition of enzymes involved in the pathology of CNS tumors. The medicinal chemistry/pharmacological activity of morpholine derivatives is discussed, in the effort to highlight the importance of morpholine ring interactions in the active site of different targets, particularly reporting binding features retrieved from PDB data, when available.
Keywords: Alzheimer’s disease; Parkinson’s disease; blood-brain barrier; brain; cancer; enzyme inhibitor.
Conflict of interest statement
The authors declare no competing financial interest.
Figures

Similar articles
-
Morpholine as ubiquitous pharmacophore in medicinal chemistry: Deep insight into the structure-activity relationship (SAR).Bioorg Chem. 2020 Mar;96:103578. doi: 10.1016/j.bioorg.2020.103578. Epub 2020 Jan 11. Bioorg Chem. 2020. PMID: 31978684 Review.
-
Morpholine as a privileged structure: A review on the medicinal chemistry and pharmacological activity of morpholine containing bioactive molecules.Med Res Rev. 2020 Mar;40(2):709-752. doi: 10.1002/med.21634. Epub 2019 Sep 12. Med Res Rev. 2020. PMID: 31512284 Review.
-
Moving beyond rules: the development of a central nervous system multiparameter optimization (CNS MPO) approach to enable alignment of druglike properties.ACS Chem Neurosci. 2010 Jun 16;1(6):435-49. doi: 10.1021/cn100008c. Epub 2010 Mar 25. ACS Chem Neurosci. 2010. PMID: 22778837 Free PMC article.
-
Integrating in Silico and in Vitro Approaches To Predict Drug Accessibility to the Central Nervous System.Mol Pharm. 2016 May 2;13(5):1540-50. doi: 10.1021/acs.molpharmaceut.6b00031. Epub 2016 Apr 4. Mol Pharm. 2016. PMID: 27015243
-
A method to predict different mechanisms for blood-brain barrier permeability of CNS activity compounds in Chinese herbs using support vector machine.J Bioinform Comput Biol. 2016 Feb;14(1):1650005. doi: 10.1142/S0219720016500050. Epub 2015 Oct 27. J Bioinform Comput Biol. 2016. PMID: 26632324
Cited by
-
Syntheses of 2- and 3-Substituted Morpholine Congeners via Ring Opening of 2-Tosyl-1,2-Oxazetidine.J Org Chem. 2023 May 5;88(9):6182-6191. doi: 10.1021/acs.joc.3c00207. Epub 2023 Apr 26. J Org Chem. 2023. PMID: 37125664 Free PMC article.
-
Discovery of imidazo[1,2-b]pyridazine-containing TAK1 kinase inhibitors with excellent activities against multiple myeloma.RSC Med Chem. 2023 Nov 28;15(1):178-192. doi: 10.1039/d3md00415e. eCollection 2024 Jan 25. RSC Med Chem. 2023. PMID: 38283221 Free PMC article.
-
Synthesis and Characterization of Copper(II) and Nickel(II) Complexes with 3-(Morpholin-4-yl)propane-2,3-dione 4-Allylthiosemicarbazone Exploring the Antibacterial, Antifungal and Antiradical Properties.Molecules. 2024 Aug 17;29(16):3903. doi: 10.3390/molecules29163903. Molecules. 2024. PMID: 39202982 Free PMC article.
-
Synthesis and In Vitro Activity of Novel Melphalan Analogs in Hematological Malignancy Cells.Int J Mol Sci. 2022 Feb 3;23(3):1760. doi: 10.3390/ijms23031760. Int J Mol Sci. 2022. PMID: 35163680 Free PMC article.
-
Short Scalable Route to Bis-morpholine Spiroacetals and Oxazepane Analogues: Useful 3D-Scaffolds for Compound Library Assembly.J Org Chem. 2025 Feb 21;90(7):2652-2661. doi: 10.1021/acs.joc.4c02690. Epub 2025 Feb 10. J Org Chem. 2025. PMID: 39927818 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

