Association of Chronic Opisthorchis Infestation and Microbiota Alteration on Tumorigenesis in Cholangiocarcinoma
- PMID: 33464733
- PMCID: PMC8345922
- DOI: 10.14309/ctg.0000000000000292
Association of Chronic Opisthorchis Infestation and Microbiota Alteration on Tumorigenesis in Cholangiocarcinoma
Abstract
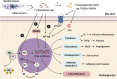
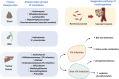
Cholangiocarcinoma (CCA) is a common hepatobiliary cancer in East and Southeast Asia. The data of microbiota contribution in CCA are still unclear. Current available reports have demonstrated that an Opisthorchis viverrini (OV) infection leads to dysbiosis in the bile duct. An increase in the commensal bacteria Helicobacter spp. in OV-infected CCA patients is associated with bile duct inflammation, severity of bile duct fibrosis, and cholangiocyte proliferation. In addition, secondary bile acids, major microbial metabolites, can mediate cholangiocyte inflammation and proliferation in the liver. A range of samples from CCA patients (stool, bile, and tumor) showed different degrees of dysbiosis. The evidence from these samples suggests that OV infection is associated with alterations in microbiota and could potentially have a role in CCA. In this comprehensive review, reports from in vitro, in vivo, and clinical studies that demonstrate possible links between OV infection, microbiota, and CCA pathogenesis are summarized and discussed. Understanding these associations may pave ways for novel potential adjunct intervention in gut microbiota in CCA patients.
Copyright © 2020 The Author(s). Published by Wolters Kluwer Health, Inc. on behalf of The American College of Gastroenterology.
Conflict of interest statement
Figures

References
-
- Bray F, Ferlay J, Soerjomataram I, et al. . Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 2018;68(6):394–424. - PubMed
-
- Squadroni M, Tondulli L, Gatta G, et al. . Cholangiocarcinoma. Crit Rev Oncol Hematol 2017;116:11–31. - PubMed
-
- Shaib Y, El-Serag HB. The epidemiology of cholangiocarcinoma. Semin Liver Dis 2004;24(2):115–25. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

