Anti-Warburg Effect of Melatonin: A Proposed Mechanism to Explain its Inhibition of Multiple Diseases
- PMID: 33466614
- PMCID: PMC7828708
- DOI: 10.3390/ijms22020764
Anti-Warburg Effect of Melatonin: A Proposed Mechanism to Explain its Inhibition of Multiple Diseases
Abstract
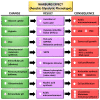
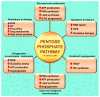
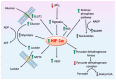
Glucose is an essential nutrient for every cell but its metabolic fate depends on cellular phenotype. Normally, the product of cytosolic glycolysis, pyruvate, is transported into mitochondria and irreversibly converted to acetyl coenzyme A by pyruvate dehydrogenase complex (PDC). In some pathological cells, however, pyruvate transport into the mitochondria is blocked due to the inhibition of PDC by pyruvate dehydrogenase kinase. This altered metabolism is referred to as aerobic glycolysis (Warburg effect) and is common in solid tumors and in other pathological cells. Switching from mitochondrial oxidative phosphorylation to aerobic glycolysis provides diseased cells with advantages because of the rapid production of ATP and the activation of pentose phosphate pathway (PPP) which provides nucleotides required for elevated cellular metabolism. Molecules, called glycolytics, inhibit aerobic glycolysis and convert cells to a healthier phenotype. Glycolytics often function by inhibiting hypoxia-inducible factor-1α leading to PDC disinhibition allowing for intramitochondrial conversion of pyruvate into acetyl coenzyme A. Melatonin is a glycolytic which converts diseased cells to the healthier phenotype. Herein we propose that melatonin's function as a glycolytic explains its actions in inhibiting a variety of diseases. Thus, the common denominator is melatonin's action in switching the metabolic phenotype of cells.
Keywords: aerobic glycolysis; hypoxia-inducible factor 1α; mitochondrial melatonin synthesis; pentose phosphate pathway; pyruvate dehydrogenase complex; pyruvate dehydrogenase kinase.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
Melatonin and Pathological Cell Interactions: Mitochondrial Glucose Processing in Cancer Cells.Int J Mol Sci. 2021 Nov 19;22(22):12494. doi: 10.3390/ijms222212494. Int J Mol Sci. 2021. PMID: 34830375 Free PMC article. Review.
-
Function of intramitochondrial melatonin and its association with Warburg metabolism.Cell Signal. 2025 Jul;131:111754. doi: 10.1016/j.cellsig.2025.111754. Epub 2025 Mar 21. Cell Signal. 2025. PMID: 40122433 Review.
-
Melatonin inhibits Warburg-dependent cancer by redirecting glucose oxidation to the mitochondria: a mechanistic hypothesis.Cell Mol Life Sci. 2020 Jul;77(13):2527-2542. doi: 10.1007/s00018-019-03438-1. Epub 2020 Jan 22. Cell Mol Life Sci. 2020. PMID: 31970423 Free PMC article. Review.
-
Melatonin synthesis in and uptake by mitochondria: implications for diseased cells with dysfunctional mitochondria.Future Med Chem. 2021 Feb;13(4):335-339. doi: 10.4155/fmc-2020-0326. Epub 2021 Jan 5. Future Med Chem. 2021. PMID: 33399498 No abstract available.
-
Switching diseased cells from cytosolic aerobic glycolysis to mitochondrial oxidative phosphorylation: A metabolic rhythm regulated by melatonin?J Pineal Res. 2021 Jan;70(1):e12677. doi: 10.1111/jpi.12677. Epub 2020 Oct 24. J Pineal Res. 2021. PMID: 32621295
Cited by
-
Molecular Research on Mitochondrial Dysfunction.Int J Mol Sci. 2022 Jun 20;23(12):6845. doi: 10.3390/ijms23126845. Int J Mol Sci. 2022. PMID: 35743286 Free PMC article.
-
Melatonin and Pathological Cell Interactions: Mitochondrial Glucose Processing in Cancer Cells.Int J Mol Sci. 2021 Nov 19;22(22):12494. doi: 10.3390/ijms222212494. Int J Mol Sci. 2021. PMID: 34830375 Free PMC article. Review.
-
Intrinsically synthesized melatonin in mitochondria and factors controlling its production.Histol Histopathol. 2025 Mar;40(3):271-282. doi: 10.14670/HH-18-776. Epub 2024 Jun 7. Histol Histopathol. 2025. PMID: 38920277 Review.
-
Crosstalk between copper, Alzheimer's disease, and melatonin.Biometals. 2025 Jul 12. doi: 10.1007/s10534-025-00712-7. Online ahead of print. Biometals. 2025. PMID: 40650773 Review.
-
Understanding the Mechanism of Action of Melatonin, Which Induces ROS Production in Cancer Cells.Antioxidants (Basel). 2022 Aug 20;11(8):1621. doi: 10.3390/antiox11081621. Antioxidants (Basel). 2022. PMID: 36009340 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

