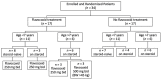
A Phase 1/2 Study of Flavocoxid, an Oral NF-κB Inhibitor, in Duchenne Muscular Dystrophy
- PMID: 33467104
- PMCID: PMC7830560
- DOI: 10.3390/brainsci11010115
A Phase 1/2 Study of Flavocoxid, an Oral NF-κB Inhibitor, in Duchenne Muscular Dystrophy
Abstract
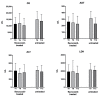
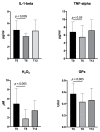
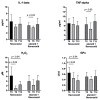
Flavocoxid is a blended extract containing baicalin and catechin with potent antioxidant and anti-inflammatory properties due to the inhibition of the cyclooxygenase (COX) and 5-lipoxygenase (5-LOX) enzymes, nuclear factor-κB (NF-κB), tumor necrosis factor (TNF)-alpha, and the mitogen-activated protein kinases (MAPKs) pathways. This phase 1/2 study was designed to assess the safety and tolerability of flavocoxid in patients with Duchenne muscular dystrophy (DMD). Thirty-four patients were recruited: 17 were treated with flavocoxid at an oral dose of 250 or 500 mg, according to body weight, for one year; 17 did not receive flavocoxid and served as controls. The treatment was well tolerated and nobody dropped out. Flavocoxid induced a significant reduction in serum interleukin (IL)-1 beta and TNF-alpha only in the group of DMD boys on add-on therapy (flavocoxid added to steroids for at least six months). The decrease in IL-1 beta was higher in younger boys. The serum H2O2 concentrations significantly decreased in patients treated with flavocoxid alone with a secondary reduction of serum glutathione peroxidase (GPx) levels, especially in younger boys. The exploratory outcome measures failed to show significant effects but there was a trend showing that the younger boys who received treatment were faster at performing the Gowers' maneuver, while the older boys who received treatment were faster at doing the 10-m walk test (10MWT). Therefore, a double-blind, placebo-controlled study for at least two/three years is warranted to verify flavocoxid as a steroid substitute or as add-on therapy to steroids.
Keywords: Duchenne muscular dystrophy; NF-κB inhibitor; anti-inflammatory agent; antioxidant; flavocoxid; phase 1/2 study.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Flavocoxid counteracts muscle necrosis and improves functional properties in mdx mice: a comparison study with methylprednisolone.Exp Neurol. 2009 Dec;220(2):349-58. doi: 10.1016/j.expneurol.2009.09.015. Epub 2009 Sep 25. Exp Neurol. 2009. PMID: 19786019
-
Antioxidant and anti-inflammatory effects of flavocoxid in high-cholesterol-fed rabbits.Naunyn Schmiedebergs Arch Pharmacol. 2015 Dec;388(12):1333-44. doi: 10.1007/s00210-015-1168-4. Epub 2015 Sep 4. Naunyn Schmiedebergs Arch Pharmacol. 2015. PMID: 26341793
-
Use of a balanced dual cyclooxygenase-1/2 and 5-lypoxygenase inhibitor in experimental colitis.Eur J Pharmacol. 2016 Oct 15;789:152-162. doi: 10.1016/j.ejphar.2016.07.033. Epub 2016 Jul 21. Eur J Pharmacol. 2016. PMID: 27450484
-
Flavocoxid, a nutraceutical approach to blunt inflammatory conditions.Mediators Inflamm. 2014;2014:790851. doi: 10.1155/2014/790851. Epub 2014 Aug 24. Mediators Inflamm. 2014. PMID: 25242871 Free PMC article. Review.
-
Natural products, PGC-1 α , and Duchenne muscular dystrophy.Acta Pharm Sin B. 2020 May;10(5):734-745. doi: 10.1016/j.apsb.2020.01.001. Epub 2020 Jan 8. Acta Pharm Sin B. 2020. PMID: 32528825 Free PMC article. Review.
Cited by
-
Current Pharmacological Strategies for Duchenne Muscular Dystrophy.Front Cell Dev Biol. 2021 Aug 19;9:689533. doi: 10.3389/fcell.2021.689533. eCollection 2021. Front Cell Dev Biol. 2021. PMID: 34490244 Free PMC article. Review.
-
New Trends to Treat Muscular Atrophy: A Systematic Review of Epicatechin.Nutrients. 2024 Jan 22;16(2):326. doi: 10.3390/nu16020326. Nutrients. 2024. PMID: 38276564 Free PMC article.
-
Oxidative Stress, Inflammation and Connexin Hemichannels in Muscular Dystrophies.Biomedicines. 2022 Feb 21;10(2):507. doi: 10.3390/biomedicines10020507. Biomedicines. 2022. PMID: 35203715 Free PMC article. Review.
-
A Randomized, Double-Blind, Placebo-Controlled, Global Phase 3 Study of Edasalonexent in Pediatric Patients with Duchenne Muscular Dystrophy: Results of the PolarisDMD Trial.J Neuromuscul Dis. 2021;8(5):769-784. doi: 10.3233/JND-210689. J Neuromuscul Dis. 2021. PMID: 34120912 Free PMC article. Clinical Trial.
-
Pharmacotherapeutic Approaches to Treatment of Muscular Dystrophies.Biomolecules. 2023 Oct 17;13(10):1536. doi: 10.3390/biom13101536. Biomolecules. 2023. PMID: 37892218 Free PMC article. Review.
References
-
- Mah J.K. An Overview of Recent Therapeutics Advances for Duchenne Muscular Dystrophy. Methods Mol. Biol. 2018;1687:3–17. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

