Glutathione in Protein Redox Modulation through S-Glutathionylation and S-Nitrosylation
- PMID: 33467703
- PMCID: PMC7838997
- DOI: 10.3390/molecules26020435
Glutathione in Protein Redox Modulation through S-Glutathionylation and S-Nitrosylation
Abstract
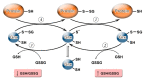
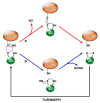
S-glutathionylation and S-nitrosylation are reversible post-translational modifications on the cysteine thiol groups of proteins, which occur in cells under physiological conditions and oxidative/nitrosative stress both spontaneously and enzymatically. They are important for the regulation of the functional activity of proteins and intracellular processes. Connecting link and "switch" functions between S-glutathionylation and S-nitrosylation may be performed by GSNO, the generation of which depends on the GSH content, the GSH/GSSG ratio, and the cellular redox state. An important role in the regulation of these processes is played by Trx family enzymes (Trx, Grx, PDI), the activity of which is determined by the cellular redox status and depends on the GSH/GSSG ratio. In this review, we analyze data concerning the role of GSH/GSSG in the modulation of S-glutathionylation and S-nitrosylation and their relationship for the maintenance of cell viability.
Keywords: GSH; S-glutathionylation; S-nitrosylation; nitrosoglutathione; redox-regulation.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Redox modulation of endothelial nitric oxide synthase by glutaredoxin-1 through reversible oxidative post-translational modification.Biochemistry. 2013 Sep 24;52(38):6712-23. doi: 10.1021/bi400404s. Epub 2013 Sep 11. Biochemistry. 2013. PMID: 23977830 Free PMC article.
-
The redox pathway of S-nitrosoglutathione, glutathione and nitric oxide in cell to neuron communications.Free Radic Res. 1999 Dec;31(6):641-50. doi: 10.1080/10715769900301211. Free Radic Res. 1999. PMID: 10630687 Review.
-
Glutathione and Glutaredoxin-Key Players in Cellular Redox Homeostasis and Signaling.Antioxidants (Basel). 2023 Aug 3;12(8):1553. doi: 10.3390/antiox12081553. Antioxidants (Basel). 2023. PMID: 37627548 Free PMC article. Review.
-
Glutaredoxin 2 catalyzes the reversible oxidation and glutathionylation of mitochondrial membrane thiol proteins: implications for mitochondrial redox regulation and antioxidant DEFENSE.J Biol Chem. 2004 Nov 12;279(46):47939-51. doi: 10.1074/jbc.M408011200. Epub 2004 Aug 30. J Biol Chem. 2004. PMID: 15347644
-
Redox Regulation by Protein S-Glutathionylation: From Molecular Mechanisms to Implications in Health and Disease.Int J Mol Sci. 2020 Oct 30;21(21):8113. doi: 10.3390/ijms21218113. Int J Mol Sci. 2020. PMID: 33143095 Free PMC article. Review.
Cited by
-
Protein Oxidative Modifications in Neurodegenerative Diseases: From Advances in Detection and Modelling to Their Use as Disease Biomarkers.Antioxidants (Basel). 2024 May 31;13(6):681. doi: 10.3390/antiox13060681. Antioxidants (Basel). 2024. PMID: 38929122 Free PMC article. Review.
-
Clinical Factors of Blood Transfusion-Related Acute Lung Injury and Changes in Levels of Treg-Related Cytokines.Emerg Med Int. 2022 May 27;2022:7344375. doi: 10.1155/2022/7344375. eCollection 2022. Emerg Med Int. 2022. Retraction in: Emerg Med Int. 2024 Jan 24;2024:9796183. doi: 10.1155/2024/9796183. PMID: 35669166 Free PMC article. Retracted.
-
Effects of short term dietary zinc deficiency and zinc supplementation on nitro-oxidative stress in testes of Wistar rats.Biometals. 2025 Apr;38(2):587-596. doi: 10.1007/s10534-025-00666-w. Epub 2025 Feb 10. Biometals. 2025. PMID: 39930252
-
Focus on Nitric Oxide Homeostasis: Direct and Indirect Enzymatic Regulation of Protein Denitrosation Reactions in Plants.Antioxidants (Basel). 2022 Jul 21;11(7):1411. doi: 10.3390/antiox11071411. Antioxidants (Basel). 2022. PMID: 35883902 Free PMC article. Review.
-
Exploring the precision redox map during fasting-refeeding and satiation in C. elegans.Stress Biol. 2023 Jun 12;3(1):17. doi: 10.1007/s44154-023-00096-z. Stress Biol. 2023. PMID: 37676352 Free PMC article.
References
-
- Giustarini D., Colombo G., Garavaglia M.L., Astori E., Portinaro N.M., Reggiani F., Badalamenti S., Aloisi A.M., Santucci A., Rossi R., et al. Assessment of glutathione/glutathione disulphide ratio and S-glutathionylated proteins in human blood, solid tissues, and cultured cells. Free Radic. Biol. Med. 2017;112:360–375. doi: 10.1016/j.freeradbiomed.2017.08.008. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

