Production of biologically active human interleukin-10 by Bifidobacterium bifidum BGN4
- PMID: 33468130
- PMCID: PMC7814708
- DOI: 10.1186/s12934-020-01505-y
Production of biologically active human interleukin-10 by Bifidobacterium bifidum BGN4
Abstract
Background: Bifidobacterium spp. are representative probiotics that play an important role in the health of their hosts. Among various Bifidobacterium spp., B. bifidum BGN4 exhibits relatively high cell adhesion to colonic cells and has been reported to have various in vivo and in vitro bio functionalities (e.g., anti-allergic effect, anti-cancer effect, and modulatory effects on immune cells). Interleukin-10 (IL-10) has emerged as a major suppressor of immune response in macrophages and other antigen presenting cells and plays an essential role in the regulation and resolution of inflammation. In this study, recombinant B. bifidum BGN4 [pBESIL10] was developed to deliver human IL-10 effectively to the intestines.
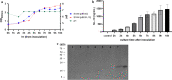
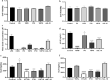
Results: The vector pBESIL10 was constructed by cloning the human IL-10 gene under a gap promoter and signal peptide from Bifidobacterium spp. into the E. coli-Bifidobacterium shuttle vector pBES2. The secreted human IL-10 from B. bifidum BGN4 [pBESIL10] was analyzed by sodium dodecyl sulphate-polyacrylamide gel electrophoresis (SDS-PAGE), Western Blotting, and enzyme-linked immunosorbent assay (ELISA). More than 1,473 ± 300 ng/mL (n = 4) of human IL-10 was obtained in the cell free culture supernatant of B. bifidum BGN4 [pBESIL10]. This productivity is significantly higher than other previously reported human IL-10 level from food grade bacteria. In vitro functional evaluation of the cell free culture supernatant of B. bifidum BGN4 [pBESIL10] revealed significantly inhibited interleukin-6 (IL-6) production in lipopolysaccharide (LPS)-induced Raw 264.7 cells (n = 6, p < 0.0001) and interleukin-8 (IL-8) production in LPS-induced HT-29 cells (n = 6, p < 0.01) or TNFα-induced HT-29 cells (n = 6, p < 0.001).
Conclusion: B. bifidum BGN4 [pBESIL10] efficiently produces and secretes significant amounts of biologically active human IL-10. The human IL-10 production level in this study is the highest of all human IL-10 production reported to date. Further research should be pursued to evaluate B. bifidum BGN4 [pBESIL10] producing IL-10 as a treatment for various inflammation-related diseases, including inflammatory bowel disease, rheumatoid arthritis, allergic asthma, and cancer immunotherapy.
Keywords: Bifidobacterium bifidum; Bioactive; Expression vector; Human interleukin-10; Recombinant; Secretion.
Conflict of interest statement
Nayoun Hong and Seockmo Ku and Tony V. Johnston declare no conflicts of interest. Kyungjin Yuk, Geun Eog Ji and Myeong Soo Park are directly employed by BIFIDO Co., Ltd. Geun Eog Ji and Myeong Soo Park hold BIFIDO Co., Ltd. stocks as a CEO and CTO, respectively.
Figures


Similar articles
-
A recombinant Bifidobacterium bifidum BGN4 strain expressing the streptococcal superoxide dismutase gene ameliorates inflammatory bowel disease.Microb Cell Fact. 2022 Jun 7;21(1):113. doi: 10.1186/s12934-022-01840-2. Microb Cell Fact. 2022. PMID: 35672695 Free PMC article.
-
Production of Selenomethionine-Enriched Bifidobacterium bifidum BGN4 via Sodium Selenite Biocatalysis.Molecules. 2018 Nov 2;23(11):2860. doi: 10.3390/molecules23112860. Molecules. 2018. PMID: 30400218 Free PMC article.
-
Safety Evaluations of Bifidobacterium bifidum BGN4 and Bifidobacterium longum BORI.Int J Mol Sci. 2018 May 9;19(5):1422. doi: 10.3390/ijms19051422. Int J Mol Sci. 2018. PMID: 29747442 Free PMC article.
-
Bifidobacterium bifidum BGN4 Paraprobiotic Supplementation Alleviates Experimental Colitis by Maintaining Gut Barrier and Suppressing Nuclear Factor Kappa B Activation Signaling Molecules.J Med Food. 2022 Feb;25(2):146-157. doi: 10.1089/jmf.2021.K.0150. J Med Food. 2022. PMID: 35148194
-
Review on Bifidobacterium bifidum BGN4: Functionality and Nutraceutical Applications as a Probiotic Microorganism.Int J Mol Sci. 2016 Sep 14;17(9):1544. doi: 10.3390/ijms17091544. Int J Mol Sci. 2016. PMID: 27649150 Free PMC article. Review.
Cited by
-
Updated Insights into Probiotic Interventions for Metabolic Syndrome: Mechanisms and Evidence.Probiotics Antimicrob Proteins. 2025 May 7. doi: 10.1007/s12602-025-10554-x. Online ahead of print. Probiotics Antimicrob Proteins. 2025. PMID: 40332670 Review.
-
Recombinant Bifidobacterium longum Carrying Endostatin Protein Alleviates Dextran Sodium Sulfate-Induced Colitis and Colon Cancer in Rats.Front Microbiol. 2022 Jun 30;13:927277. doi: 10.3389/fmicb.2022.927277. eCollection 2022. Front Microbiol. 2022. PMID: 35847065 Free PMC article.
-
Modulation of Gut Microbiota for the Prevention and Treatment of COVID-19.J Clin Med. 2021 Jun 29;10(13):2903. doi: 10.3390/jcm10132903. J Clin Med. 2021. PMID: 34209870 Free PMC article. Review.
-
Antioxidant and Anti-Inflammatory Properties of Recombinant Bifidobacterium bifidum BGN4 Expressing Antioxidant Enzymes.Microorganisms. 2021 Mar 13;9(3):595. doi: 10.3390/microorganisms9030595. Microorganisms. 2021. PMID: 33805797 Free PMC article.
-
A recombinant Bifidobacterium bifidum BGN4 strain expressing the streptococcal superoxide dismutase gene ameliorates inflammatory bowel disease.Microb Cell Fact. 2022 Jun 7;21(1):113. doi: 10.1186/s12934-022-01840-2. Microb Cell Fact. 2022. PMID: 35672695 Free PMC article.
References
-
- Biavati B, Mattarelli P. Bifidobacterium Bergey's Manual of systematics of archaea and bacteria 2015; 1–57.
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

