Somatostatin Neurons of the Bed Nucleus of Stria Terminalis Enhance Associative Fear Memory Consolidation in Mice
- PMID: 33468566
- PMCID: PMC7939080
- DOI: 10.1523/JNEUROSCI.1944-20.2020
Somatostatin Neurons of the Bed Nucleus of Stria Terminalis Enhance Associative Fear Memory Consolidation in Mice
Abstract
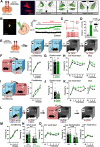
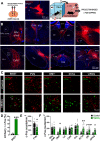
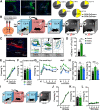
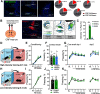
Excessive fear learning and generalized, extinction-resistant fear memories are core symptoms of anxiety and trauma-related disorders. Despite significant evidence from clinical studies reporting hyperactivity of the bed nucleus of stria terminalis (BNST) under these conditions, the role of BNST in fear learning and expression is still not clarified. Here, we tested how BNST modulates fear learning in male mice using a chemogenetic approach. Activation of GABAergic neurons of BNST during fear conditioning or memory consolidation resulted in enhanced cue-related fear recall. Importantly, BNST activation had no acute impact on fear expression during conditioning or recalls, but it enhanced cue-related fear recall subsequently, potentially via altered activity of downstream regions. Enhanced fear memory consolidation could be replicated by selectively activating somatostatin (SOM), but not corticotropin-releasing factor (CRF), neurons of the BNST, which was accompanied by increased fear generalization. Our findings suggest the significant modulation of fear memory strength by specific circuits of the BNST.SIGNIFICANCE STATEMENT The bed nucleus of stria terminalis (BNST) mediates different defensive behaviors, and its connections implicate its integrative modulatory role in fear memory formation; however, the involvement of BNST in fear learning has yet to be elucidated in detail. Our data highlight that BNST stimulation enhances fear memory formation without direct effects on fear expression. Our study identified somatostatin (SOM) cells within the extended amygdala as specific neurons promoting fear memory formation. These data underline the importance of anxiety circuits in maladaptive fear memory formation, indicating elevated BNST activity as a potential vulnerability factor to anxiety and trauma-related disorders.
Keywords: BNST; DREADD; extended amygdala; fear; somatostatin..
Copyright © 2021 the authors.
Figures


Similar articles
-
A prefrontal-bed nucleus of the stria terminalis circuit limits fear to uncertain threat.Elife. 2020 Dec 15;9:e60812. doi: 10.7554/eLife.60812. Elife. 2020. PMID: 33319747 Free PMC article.
-
A Central Extended Amygdala Circuit That Modulates Anxiety.J Neurosci. 2018 Jun 13;38(24):5567-5583. doi: 10.1523/JNEUROSCI.0705-18.2018. Epub 2018 May 29. J Neurosci. 2018. PMID: 29844022 Free PMC article.
-
Chronic Stress Induces Maladaptive Behaviors by Activating Corticotropin-Releasing Hormone Signaling in the Mouse Oval Bed Nucleus of the Stria Terminalis.J Neurosci. 2020 Mar 18;40(12):2519-2537. doi: 10.1523/JNEUROSCI.2410-19.2020. Epub 2020 Feb 13. J Neurosci. 2020. PMID: 32054675 Free PMC article.
-
Chemoarchitecture of the bed nucleus of the stria terminalis: Neurophenotypic diversity and function.Handb Clin Neurol. 2021;179:385-402. doi: 10.1016/B978-0-12-819975-6.00025-X. Handb Clin Neurol. 2021. PMID: 34225977 Free PMC article. Review.
-
Role of the bed nucleus of the stria terminalis in aversive learning and memory.Learn Mem. 2017 Aug 16;24(9):480-491. doi: 10.1101/lm.044206.116. Print 2017 Sep. Learn Mem. 2017. PMID: 28814474 Free PMC article. Review.
Cited by
-
Sex differences in distribution and identity of aromatase gene expressing cells in the young adult rat brain.Biol Sex Differ. 2023 Sep 1;14(1):54. doi: 10.1186/s13293-023-00541-8. Biol Sex Differ. 2023. PMID: 37658400 Free PMC article.
-
Serotonergic modulation of the BNST-CeA pathway reveals sex differences in fear learning.Nat Neurosci. 2025 Aug 6. doi: 10.1038/s41593-025-02025-x. Online ahead of print. Nat Neurosci. 2025. PMID: 40770508
-
Sex differences in neural projections of fear memory processing in mice and humans.Sci Adv. 2024 Jul 12;10(28):eadk3365. doi: 10.1126/sciadv.adk3365. Epub 2024 Jul 10. Sci Adv. 2024. PMID: 38985873 Free PMC article.
-
Growth hormone and IGF-1 actions in the brain and neuropsychiatric diseases.Physiology (Bethesda). 2025 Jul 7:10.1152/physiol.00009.2025. doi: 10.1152/physiol.00009.2025. Online ahead of print. Physiology (Bethesda). 2025. PMID: 40623083 Free PMC article. Review.
-
Ovariectomy-induced hormone deprivation aggravates Aβ1-42 deposition in the basolateral amygdala and cholinergic fiber loss in the cortex but not cognitive behavioral symptoms in a triple transgenic mouse model of Alzheimer's disease.Front Endocrinol (Lausanne). 2022 Oct 11;13:985424. doi: 10.3389/fendo.2022.985424. eCollection 2022. Front Endocrinol (Lausanne). 2022. PMID: 36303870 Free PMC article.
References
-
- Brinkmann L, Buff C, Feldker K, Tupak SV, Becker MPI, Herrmann MJ, Straube T (2017) Distinct phasic and sustained brain responses and connectivity of amygdala and bed nucleus of the stria terminalis during threat anticipation in panic disorder. Psychol Med 47:2675–2688. 10.1017/S0033291717001192 - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
