Carboxypeptidase U (CPU, TAFIa, CPB2) in Thromboembolic Disease: What Do We Know Three Decades after Its Discovery?
- PMID: 33477318
- PMCID: PMC7830380
- DOI: 10.3390/ijms22020883
Carboxypeptidase U (CPU, TAFIa, CPB2) in Thromboembolic Disease: What Do We Know Three Decades after Its Discovery?
Abstract
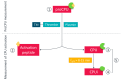
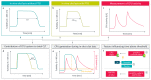
Procarboxypeptidase U (proCPU, TAFI, proCPB2) is a basic carboxypeptidase zymogen that is converted by thrombin(-thrombomodulin) or plasmin into the active carboxypeptidase U (CPU, TAFIa, CPB2), a potent attenuator of fibrinolysis. As CPU forms a molecular link between coagulation and fibrinolysis, the development of CPU inhibitors as profibrinolytic agents constitutes an attractive new concept to improve endogenous fibrinolysis or to increase the efficacy of thrombolytic therapy in thromboembolic diseases. Furthermore, extensive research has been conducted on the in vivo role of CPU in (the acute phase of) thromboembolic disease, as well as on the hypothesis that high proCPU levels and the Thr/Ile325 polymorphism may cause a thrombotic predisposition. In this paper, an overview is given of the methods available for measuring proCPU, CPU, and inactivated CPU (CPUi), together with a summary of the clinical data generated so far, ranging from the current knowledge on proCPU concentrations and polymorphisms as potential thromboembolic risk factors to the positioning of different CPU forms (proCPU, CPU, and CPUi) as diagnostic markers for thromboembolic disease, and the potential benefit of pharmacological inhibition of the CPU pathway.
Keywords: arterial thrombosis; carboxypeptidase B2; carboxypeptidase U; fibrinolysis; thrombin-activatable fibrinolysis inhibitor; thromboembolic diseases; venous thrombosis.
Conflict of interest statement
D. Hendriks reports consultancy and services for the Institut de Recherches Internationales Servier. The other authors declare no conflict of interest.
Figures

Similar articles
-
Plasma carboxypeptidase U (CPU, CPB2, TAFIa) generation during in vitro clot lysis and its interplay between coagulation and fibrinolysis.Thromb Haemost. 2017 Jul 26;117(8):1498-1508. doi: 10.1160/TH17-02-0097. Epub 2017 Jul 6. Thromb Haemost. 2017. PMID: 28692110
-
Activation of the Carboxypeptidase U (CPU, TAFIa, CPB2) System in Patients with SARS-CoV-2 Infection Could Contribute to COVID-19 Hypofibrinolytic State and Disease Severity Prognosis.J Clin Med. 2022 Mar 9;11(6):1494. doi: 10.3390/jcm11061494. J Clin Med. 2022. PMID: 35329820 Free PMC article.
-
Plasma levels of carboxypeptidase U (CPU, CPB2 or TAFIa) are elevated in patients with acute myocardial infarction.J Thromb Haemost. 2015 Dec;13(12):2227-32. doi: 10.1111/jth.13135. Epub 2015 Nov 12. J Thromb Haemost. 2015. PMID: 26340515
-
An update on the role of carboxypeptidase U (TAFIa) in fibrinolysis.Front Biosci (Landmark Ed). 2011 Jun 1;16(7):2427-50. doi: 10.2741/3864. Front Biosci (Landmark Ed). 2011. PMID: 21622187 Review.
-
Carboxypeptidase U (TAFIa): a new drug target for fibrinolytic therapy?J Thromb Haemost. 2009 Dec;7(12):1962-71. doi: 10.1111/j.1538-7836.2009.03596.x. Epub 2009 Aug 28. J Thromb Haemost. 2009. PMID: 19719827 Free PMC article. Review.
Cited by
-
Comparison of the proteomic landscape in experimental ischemia reperfusion with versus without ischemic preconditioning.Sci Rep. 2025 Apr 7;15(1):11836. doi: 10.1038/s41598-025-90735-4. Sci Rep. 2025. PMID: 40195349 Free PMC article.
-
Efficacy and Mechanism of Buyang Huanwu Decoction in Patients With Ischemic Heart Failure: A Randomized, Double-Blind, Placebo-Controlled Trial Combined With Proteomic Analysis.Front Pharmacol. 2022 Mar 18;13:831208. doi: 10.3389/fphar.2022.831208. eCollection 2022. Front Pharmacol. 2022. PMID: 35370712 Free PMC article.
-
Pleiotropic Effects of Atorvastatin Result in a Downregulation of the Carboxypeptidase U System (CPU, TAFIa, CPB2) in a Mouse Model of Advanced Atherosclerosis.Pharmaceutics. 2021 Oct 19;13(10):1731. doi: 10.3390/pharmaceutics13101731. Pharmaceutics. 2021. PMID: 34684024 Free PMC article.
-
Recent Pathophysiological Insights Are Advancing the Treatment of Venous Thromboembolism.JACC Basic Transl Sci. 2025 May;10(5):689-703. doi: 10.1016/j.jacbts.2024.12.004. Epub 2025 Feb 26. JACC Basic Transl Sci. 2025. PMID: 40436530 Free PMC article. Review.
-
Thrombin Activatable Fibrinolysis Inhibitor (TAFI): An Updated Narrative Review.Int J Mol Sci. 2021 Apr 1;22(7):3670. doi: 10.3390/ijms22073670. Int J Mol Sci. 2021. PMID: 33916027 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

