Chemical Diversity and Bioactivities of Monoterpene Indole Alkaloids (MIAs) from Six Apocynaceae Genera
- PMID: 33477682
- PMCID: PMC7831967
- DOI: 10.3390/molecules26020488
Chemical Diversity and Bioactivities of Monoterpene Indole Alkaloids (MIAs) from Six Apocynaceae Genera
Abstract
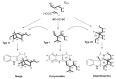
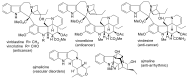
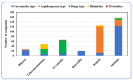
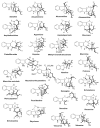
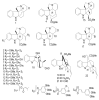
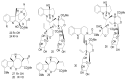
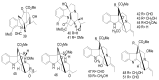
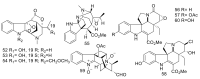
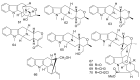
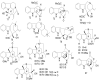
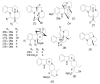
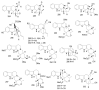
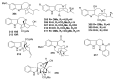
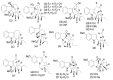
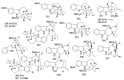
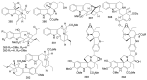
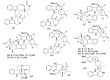
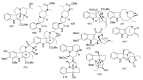
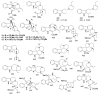
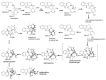
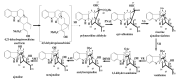
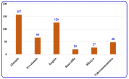
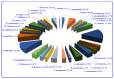
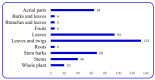
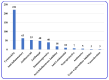
By the end of the twentieth century, the interest in natural compounds as probable sources of drugs has declined and was replaced by other strategies such as molecular target-based drug discovery. However, in the recent times, natural compounds regained their position as extremely important source drug leads. Indole-containing compounds are under clinical use which includes vinblastine and vincristine (anticancer), atevirdine (anti-HIV), yohimbine (erectile dysfunction), reserpine (antihypertension), ajmalicine (vascular disorders), ajmaline (anti-arrhythmic), vincamine (vasodilator), etc. Monoterpene Indole Alkaloids (MIAs) deserve the curiosity and attention of researchers due to their chemical diversity and biological activities. These compounds were considered as an impending source of drug-lead. In this review 444 compounds, were identified from six genera belonging to the family Apocynaceae, will be discussed. These genera (Alstonia, Rauvolfia, Kopsia, Ervatamia, and Tabernaemontana, and Rhazya) consist of 400 members and represent 20% of Apocynaceae species. Only 30 (7.5%) species were investigated, whereas the rest are promising to be investigated. Eleven bioactivities, including antibacterial, antifungal, anti-inflammatory and immunosuppressant activities, were reported. Whereas cytotoxic effect represents 47% of the reported activities. Convincingly, the genera selected in this review are a wealthy source for future anticancer drug lead.
Keywords: Apocynaceae; alkaloids; anti-inflammatory; antimicrobial; cytotoxicity; monoterpene.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
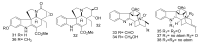
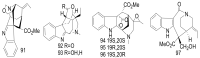
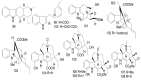
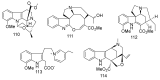
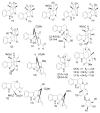
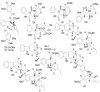
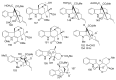
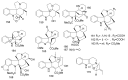
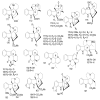
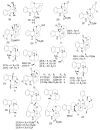




References
-
- Dey A., Mukherjee A., Chaudhury M. Alkaloids from Apocynaceae: Origin, pharmacotherapeutic properties, and structure-activity studies. Stud. Nat. Prod. Chem. 2017;52:373–488.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

