Computational and theoretical insights into the homeostatic response to the decreased cell size of midbrain dopamine neurons
- PMID: 33484235
- PMCID: PMC7824968
- DOI: 10.14814/phy2.14709
Computational and theoretical insights into the homeostatic response to the decreased cell size of midbrain dopamine neurons
Abstract
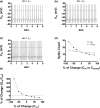
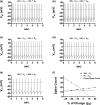
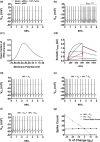
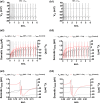
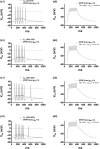
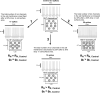
Midbrain dopamine neurons communicate signals of reward anticipation and attribution of salience. This capacity is distorted in heroin or cocaine abuse or in conditions such as human mania. A shared characteristic among rodent models of these behavioral disorders is that dopamine neurons in these animals acquired a small size and manifest an augmented spontaneous and burst activity. The biophysical mechanism underlying this increased excitation is currently unknown, but is believed to primarily follow from a substantial drop in K+ conductance secondary to morphology reduction. This work uses a dopamine neuron mathematical model to show, surprisingly, that under size diminution a reduction in K+ conductance is an adaptation that attempts to decrease cell excitability. The homeostatic response that preserves the intrinsic activity is the conservation of the ion channel density for each conductance; a result that is analytically demonstrated and challenges the experimentalist tendency to reduce intrinsic excitation to K+ conductance expression level. Another unexpected mechanism that buffers the raise in intrinsic activity is the presence of the ether-a-go-go-related gen K+ channel since its activation is illustrated to increase with size reduction. Computational experiments finally demonstrate that size attenuation results in the paradoxical enhancement of afferent-driven bursting as a reduced temporal summation indexed correlates with improved depolarization. This work illustrates, on the whole, that experimentation in the absence of mathematical models may lead to the erroneous interpretation of the counterintuitive aspects of empirical data.
Keywords: capacitance; cell size; computational modeling; dopamine neurons.
© 2020 The Authors. Physiological Reports published by Wiley Periodicals LLC on behalf of The Physiological Society and the American Physiological Society.
Conflict of interest statement
No conflicts of interest, financial or otherwise, are declared by the authors.
Figures

References
-
- Arencibia‐Albite, F. , Vázquez, R. , Velásquez‐Martinez, M. C. , & Jiménez‐Rivera, C. A. (2012). Cocaine sensitization inhibits the hyperpolarization‐activated cation current Ih and reduces cell size in dopamine neurons of the ventral tegmental area. Journal of Neurophysiology, 107(8), 2271–2282. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

