Partnership for Research on Ebola VACcination (PREVAC): protocol of a randomized, double-blind, placebo-controlled phase 2 clinical trial evaluating three vaccine strategies against Ebola in healthy volunteers in four West African countries
- PMID: 33485369
- PMCID: PMC7823170
- DOI: 10.1186/s13063-021-05035-9
Partnership for Research on Ebola VACcination (PREVAC): protocol of a randomized, double-blind, placebo-controlled phase 2 clinical trial evaluating three vaccine strategies against Ebola in healthy volunteers in four West African countries
Erratum in
-
Correction to: Partnership for Research on Ebola VACcination (PREVAC): protocol of a randomized, double-blind, placebo-controlled phase 2 clinical trial evaluating three vaccine strategies against Ebola in healthy volunteers in four West African countries.Trials. 2021 Sep 1;22(1):583. doi: 10.1186/s13063-021-05572-3. Trials. 2021. PMID: 34470655 Free PMC article. No abstract available.
Abstract
Introduction: The Ebola virus disease (EVD) outbreak in 2014-2016 in West Africa was the largest on record and provided an opportunity for large clinical trials and accelerated efforts to develop an effective and safe preventative vaccine. Multiple questions regarding the safety, immunogenicity, and efficacy of EVD vaccines remain unanswered. To address these gaps in the evidence base, the Partnership for Research on Ebola Vaccines (PREVAC) trial was designed. This paper describes the design, methods, and baseline results of the PREVAC trial and discusses challenges that led to different protocol amendments.
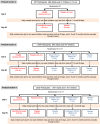
Methods: This is a randomized, double-blind, placebo-controlled phase 2 clinical trial of three vaccine strategies against the Ebola virus in healthy volunteers 1 year of age and above. The three vaccine strategies being studied are the rVSVΔG-ZEBOV-GP vaccine, with and without a booster dose at 56 days, and the Ad26.ZEBOV,MVA-FN-Filo vaccine regimen with Ad26.ZEBOV given as the first dose and the MVA-FN-Filo vaccination given 56 days later. There have been 4 versions of the protocol with those enrolled in Version 4.0 comprising the primary analysis cohort. The primary endpoint is based on the antibody titer against the Ebola virus surface glycoprotein measured 12 months following the final injection.
Results: From April 2017 to December 2018, a total of 5002 volunteers were screened and 4789 enrolled. Participants were enrolled at 6 sites in four countries (Guinea, Liberia, Sierra Leone, and Mali). Of the 4789 participants, 2560 (53%) were adults and 2229 (47%) were children. Those < 18 years of age included 549 (12%) aged 1 to 4 years, 750 (16%) 5 to 11 years, and 930 (19%) aged 12-17 years. At baseline, the median (25th, 75th percentile) antibody titer to Ebola virus glycoprotein for 1090 participants was 72 (50, 116) EU/mL.
Discussion: The PREVAC trial is evaluating-placebo-controlled-two promising Ebola candidate vaccines in advanced stages of development. The results will address unanswered questions related to short- and long-term safety and immunogenicity for three vaccine strategies in adults and children.
Trial registration: ClinicalTrials.gov NCT02876328 . Registered on 23 August 2016.
Keywords: Clinical trials; Ebola; Protocol; Randomized controlled trials; Vaccine.
Conflict of interest statement
None declared.
Figures
References
-
- Henao-Restrepo AM, Camacho A, Longini IM, et al. Efficacy and effectiveness of an rVSV-vectored vaccine in preventing Ebola virus disease: final results from the Guinea ring vaccination, open-label, cluster-randomised trial (Ebola Ça Suffit!). Lancet. 2017. 10.1016/S0140-6736(16)32621-6. - PMC - PubMed
-
- Milligan ID, Gibani MM, Sewell R, et al. Safety and immunogenicity of novel adenovirus type 26-and modified vaccinia Ankara-vectored Ebola vaccines: a randomized clinical trial. JAMA. 2016. 10.1001/jama.2016.4218. - PubMed
-
- Pollard AJ, Launay O, Lelievre J-D, et al. Safety and immunogenicity of a two-dose heterologous Ad26.ZEBOV and MVA-BN-Filo Ebola vaccine regimen in adults in Europe (EBOVAC2): a randomised, observer-blind, participant-blind, placebo-controlled, phase 2 trial. Lancet Infect Dis. 2020. 10.1016/s1473-3099(20)30476-x. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous