Selenium-Containing Amino Acids Protect Dextran Sulfate Sodium-Induced Colitis via Ameliorating Oxidative Stress and Intestinal Inflammation
- PMID: 33488110
- PMCID: PMC7814278
- DOI: 10.2147/JIR.S288412
Selenium-Containing Amino Acids Protect Dextran Sulfate Sodium-Induced Colitis via Ameliorating Oxidative Stress and Intestinal Inflammation
Abstract
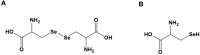
Background: Inflammatory bowel disease (IBD) is characterized by chronic relapsing inflammation of the gastrointestinal tract. Oxidative stress plays a pivotal role in the pathogenesis of IBD. Selenium-containing amino acids reportedly have anti-oxidative and anti-inflammatory properties, but it remains unknown if selenium-containing amino acids can be used to treat IBD. This study aimed to investigate the effects of two selenium-containing amino acids - selenocysteine and selenocystine - on oxidative stress and chronic inflammation in a mouse model of dextran sulfate sodium (DSS)-induced IBD.
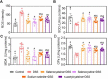
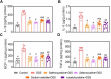
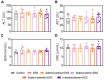
Methodology: C57BL/6 mice were randomly assigned to the following six groups: control, DSS, DSS+selenocysteine, DSS+selenocystine, DSS+sodium selenite, and DSS+N-acetylcysteine (NAC). IBD was induced by 3% DSS. Pro-inflammatory cytokines [interleukin-1β (IL-1β), monocyte chemotactic protein 1 (MCP-1), IL-6, and tumor necrosis factor-α (TNF-α)] and markers for oxidative and anti-oxidative stress [malondialdehyde (MDA), reactive oxygen species (ROS), superoxide dismutase (SOD), and glutathione peroxidase (GPx)] were measured using immunohistochemical analysis.
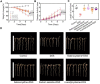
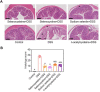
Results: Selenocysteine and selenocystine significantly attenuated IBD-related symptoms, including preventing weight loss, decreasing disease activity index (DAI) scores, and increasing colon length. Selenocysteine and selenocystine significantly ameliorated the DSS-induced oxidative stress, as demonstrated by a reduction in ROS and MDA activity and an increase in SOD and GPx activity. IL-1, MCP-1, IL-6, and TNF-α levels were significantly increased in the IBD mice, while treatment with the selenium-containing amino acids significantly reduced the levels of these pro-inflammatory cytokines. In vivo safety analysis showed minimal side effects of the selenium-containing amino acids.
Conclusion: We found that selenocysteine and selenocystine ameliorated DSS-induced IBD via reducing oxidative stress and intestinal inflammation, indicating that selenium-containing amino acids could be a novel therapeutic option for patients with IBD.
Keywords: Inflammatory bowel diseases; colitis; inflammation; oxidative stress; selenium-containing amino acids; selenocysteine; selenocystine.
© 2021 Shi et al.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Figures

References
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

