Increased Excitability and Heightened Magnitude of Long-Term Potentiation at Hippocampal CA3-CA1 Synapses in a Mouse Model of Neonatal Hyperoxia Exposure
- PMID: 33488380
- PMCID: PMC7815524
- DOI: 10.3389/fnsyn.2020.609903
Increased Excitability and Heightened Magnitude of Long-Term Potentiation at Hippocampal CA3-CA1 Synapses in a Mouse Model of Neonatal Hyperoxia Exposure
Abstract
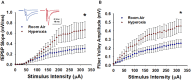
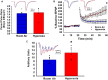
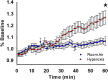
Preterm infants exposed to supraphysiological oxygen (hyperoxia) during the neonatal period have hippocampal atrophy and cognitive dysfunction later in childhood and as adolescents. Previously, we reported that 14-week-old adult mice exposed to hyperoxia as newborns had spatial memory deficits and hippocampal shrinkage, findings that mirror those of human adolescents who were born preterm. The area CA1 region of the hippocampus that is crucial for spatial learning and memory is highly vulnerable to oxidative stress. In this study, we investigated the long-term impact of neonatal hyperoxia exposure on hippocampal CA3-CA1 synaptic function. Male and female C57BL/6J mouse pups were continuously exposed to either 85% normobaric oxygen or air between postnatal days 2-14. Hippocampal slice electrophysiology at CA3-CA1 synapses was then performed at 14 weeks of age. We observed that hyperoxia exposed mice have heightened strength of basal synaptic transmission measured in input-output curves, increased fiber volley amplitude indicating increased axonal excitability, and heightened LTP magnitude at CA3-CA1 synapses, likely a consequence of increased postsynaptic depolarization during tetanus. These data demonstrate that supraphysiological oxygen exposure during the critical neonatal developmental period leads to pathologically heightened CA3-CA1 synaptic function during early adulthood which may contribute to hippocampal shrinkage and learning and memory deficits we previously reported. Furthermore, these results will help shed light on the consequences of hyperoxia exposure on the development of hippocampal synaptic circuit abnormalities that could be contributing to cognitive deficits in children born preterm.
Keywords: early-life insult; hyperoxia; long-term hippocampal dysfunction; neuronal excitability; preterm; synaptic transmission.
Copyright © 2021 Ramani, Miller, Ambalavanan and McMahon.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
References
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

