Role of Proprotein Convertase Subtilisin/Kexin Type 9 in the Pathogenesis of Graves' Orbitopathy in Orbital Fibroblasts
- PMID: 33488522
- PMCID: PMC7821242
- DOI: 10.3389/fendo.2020.607144
Role of Proprotein Convertase Subtilisin/Kexin Type 9 in the Pathogenesis of Graves' Orbitopathy in Orbital Fibroblasts
Abstract
Background: The proprotein convertase subtilisin/kexin type 9 (PCSK9) has been implicated in the pathogenesis of inflammatory diseases. We sought to investigate the role of PCSK9 in the pathogenesis of Graves' orbitopathy (GO) and whether it may be a legitimate target for treatment.
Methods: The PCSK9 was compared between GO (n=11) and normal subjects (n=7) in orbital tissue explants using quantitative real-time PCR, and in cultured interleukin-1β (IL-1β)-treated fibroblasts using western blot. Western blot was used to identify the effects of PCSK9 inhibition on IL-1β-induced pro-inflammatory cytokines production and signaling molecules expression as well as levels of adipogenic markers and oxidative stress-related proteins. Adipogenic differentiation was identified using Oil Red O staining. The plasma PCSK9 concentrations were compared between patients with GO (n=44) and healthy subjects (n=26) by ELISA.
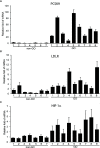
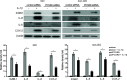
Results: The PCSK9 transcript level was higher in GO tissues. The depletion of PCSK9 blunted IL-1β-induced expression of intercellular adhesion molecule 1 (ICAM-1), IL-6, IL-8, and cyclooxygenase-2 (COX-2) in GO and non-GO fibroblasts. The levels of activated nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) and phosphorylated forms of Akt and p38 were diminished when PCSK9 was suppressed in GO fibroblasts. Decreases in lipid droplets and attenuated levels of peroxisome proliferator-activated receptor gamma (PPARγ), CCAAT/enhancer-binding protein β (C/EBPβ), and leptin as well as hypoxia-inducible factor 1α (HIF-1α), manganese superoxide dismutase (MnSOD), thioredoxin (Trx), and heme oxygenase-1 (HO-1) were noted when PCSK9 was suppressed during adipocyte differentiation. The plasma PCSK9 level was significantly higher in GO patients and correlated with level of thyrotropin binding inhibitory immunoglobulin (TBII) and the clinical activity score (CAS).
Conclusions: PCSK9 plays a significant role in GO. The PCSK9 inhibition attenuated the pro-inflammatory cytokines production, oxidative stress, and fibroblast differentiation into adipocytes. PCSK9 may serve as a therapeutic target and biomarker for GO.
Keywords: Graves’ orbitopathy; PCSK9; adipogenesis; inflammation; oxidative stress; proprotein convertase subtilisin/kexin type 9; thyroid eye disease.
Copyright © 2021 Lee, Kim, Lee, Ko, Lee and Yoon.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures




Similar articles
-
Glycogen Synthase Kinase-3β Mediates Proinflammatory Cytokine Secretion and Adipogenesis in Orbital Fibroblasts from Patients with Graves' Orbitopathy.Invest Ophthalmol Vis Sci. 2020 Jul 1;61(8):51. doi: 10.1167/iovs.61.8.51. Invest Ophthalmol Vis Sci. 2020. PMID: 32735324 Free PMC article.
-
Role of Lysyl Oxidase-Like Protein 3 in the Pathogenesis of Graves' Orbitopathy in Orbital Fibroblasts.Invest Ophthalmol Vis Sci. 2024 Nov 4;65(13):33. doi: 10.1167/iovs.65.13.33. Invest Ophthalmol Vis Sci. 2024. PMID: 39546293 Free PMC article.
-
Pentraxin 3 mediates inflammation and adipogenesis in Graves' orbitopathy pathogenesis.J Mol Endocrinol. 2024 Oct 14;73(4):e240039. doi: 10.1530/JME-24-0039. Print 2024 Nov 1. J Mol Endocrinol. 2024. PMID: 39283923
-
Current perspectives on the role of orbital fibroblasts in the pathogenesis of Graves' ophthalmopathy.Exp Eye Res. 2016 Jan;142:83-91. doi: 10.1016/j.exer.2015.02.007. Exp Eye Res. 2016. PMID: 26675405 Review.
-
Molecular biomarkers of Graves' ophthalmopathy.Exp Mol Pathol. 2019 Feb;106:1-6. doi: 10.1016/j.yexmp.2018.11.004. Epub 2018 Nov 8. Exp Mol Pathol. 2019. PMID: 30414981 Free PMC article. Review.
Cited by
-
Role of the suppressor of cytokine signaling-3 in the pathogenesis of Graves' orbitopathy.Front Endocrinol (Lausanne). 2025 Mar 4;16:1527275. doi: 10.3389/fendo.2025.1527275. eCollection 2025. Front Endocrinol (Lausanne). 2025. PMID: 40104138 Free PMC article.
-
PCSK9 Inhibition Regulates Infarction-Induced Cardiac Myofibroblast Transdifferentiation via Notch1 Signaling.Cell Biochem Biophys. 2023 Jun;81(2):359-369. doi: 10.1007/s12013-023-01136-1. Epub 2023 Apr 21. Cell Biochem Biophys. 2023. PMID: 37081375
-
Thyroid eye disease: From pathogenesis to targeted therapies.Taiwan J Ophthalmol. 2022 Jan 21;12(1):3-11. doi: 10.4103/tjo.tjo_51_21. eCollection 2022 Jan-Mar. Taiwan J Ophthalmol. 2022. PMID: 35399971 Free PMC article. Review.
-
Association of fibroblast growth factor 10 with the fibrotic and inflammatory pathogenesis of Graves' orbitopathy.PLoS One. 2021 Aug 12;16(8):e0255344. doi: 10.1371/journal.pone.0255344. eCollection 2021. PLoS One. 2021. PMID: 34383782 Free PMC article.
-
PCSK9 Enhances Cardiac Fibrogenesis via the Activation of Toll-like Receptor and NLRP3 Inflammasome Signaling.Int J Mol Sci. 2025 Feb 23;26(5):1921. doi: 10.3390/ijms26051921. Int J Mol Sci. 2025. PMID: 40076547 Free PMC article.
References
-
- Seidah NG, Benjannet S, Wickham L, Marcinkiewicz J, Jasmin SB, Stifani S, et al. The secretory proprotein convertase neural apoptosis-regulated convertase 1 (NARC-1): liver regeneration and neuronal differentiation. Proc Natl Acad Sci (2003) 100(3):928–33. 10.1073/pnas.0335507100 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

