Design, synthesis, and biological evaluation of novel 5,6,7-trimethoxy quinolines as potential anticancer agents and tubulin polymerization inhibitors
- PMID: 33489025
- PMCID: PMC7811808
- DOI: 10.22038/ijbms.2020.43303.10168
Design, synthesis, and biological evaluation of novel 5,6,7-trimethoxy quinolines as potential anticancer agents and tubulin polymerization inhibitors
Abstract
Objectives: Microtubules have key roles in essential cellular processes such as mitosis, cell motion, and intracellular organelle transport. Increasing interest has been given to tubulin binding compounds after the introduction of taxanes into clinical oncology. The object of this study was synthesis and biological evaluation of novel 5,6,7-trimethoxy quinolines as tubulin inhibitors.
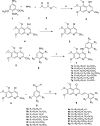
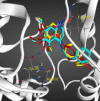
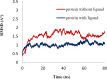
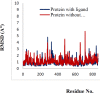
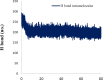
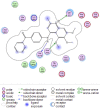
Materials and methods: The cytotoxicity of the newly synthesized compounds was assessed against different human cancer cell lines including MCF-7, A2780, MCF-7/MX, A2780/RCIS, and normal cells. Compounds demonstrating the most antiproliferative activity, were chosen to examine their tubulin inhibition activity and their ability to arrest the cell cycle and induce apoptosis. Molecular docking studies and molecular dynamics simulation of compound 7e in the catalytic site of tubulin were performed.
Results: Most of the synthesized quinolines showed moderate to significant cytotoxic activity against human cancer cells. Compounds 7e and 7f, possessing N-(4-benzoyl phenyl) and N-(4-phenoxy phenyl), respectively, exhibited the most antiproliferative activity more potent than the other compounds and exhibited similar antiproliferative activity on both resistant and parental cancer cells.
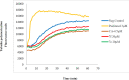
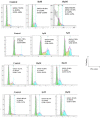
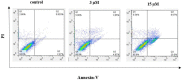
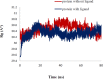
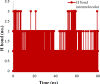
Conclusion: Flow cytometry analysis of A2780, A2780/RCIS, MCF-7, and MCF-7/MX cancer cells treated with 7e and 7f exhibited that these compounds arrested the cell cycle (at the G2/M phase) and induced cellular apoptosis in A2780 cancer cells. These quinolines inhibited tubulin polymerization in a way resembling that of CA-4. Molecular dynamics simulation and molecular docking studies of compound 7e into the binding site of tubulin displayed the probable interactions of 7e with the binding site of tubulin.
Keywords: Anticancer activity; Apoptosis; Quinoline; Resistant cancer cells; Tubulin inhibitors.
Figures

References
LinkOut - more resources
Full Text Sources
