Berberine Prevents Disease Progression of Nonalcoholic Steatohepatitis through Modulating Multiple Pathways
- PMID: 33494295
- PMCID: PMC7912096
- DOI: 10.3390/cells10020210
Berberine Prevents Disease Progression of Nonalcoholic Steatohepatitis through Modulating Multiple Pathways
Abstract
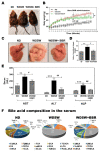
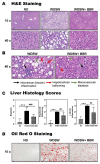
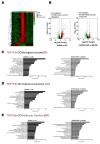
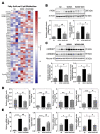
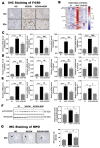
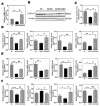
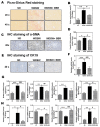
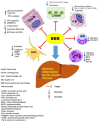
The disease progression of nonalcoholic fatty liver disease (NAFLD) from simple steatosis (NAFL) to nonalcoholic steatohepatitis (NASH) is driven by multiple factors. Berberine (BBR) is an ancient Chinese medicine and has various beneficial effects on metabolic diseases, including NAFLD/NASH. However, the underlying mechanisms remain incompletely understood due to the limitation of the NASH animal models used. Methods: A high-fat and high-fructose diet-induced mouse model of NAFLD, the best available preclinical NASH mouse model, was used. RNAseq, histological, and metabolic pathway analyses were used to identify the potential signaling pathways modulated by BBR. LC-MS was used to measure bile acid levels in the serum and liver. The real-time RT-PCR and Western blot analysis were used to validate the RNAseq data. Results: BBR not only significantly reduced hepatic lipid accumulation by modulating fatty acid synthesis and metabolism but also restored the bile acid homeostasis by targeting multiple pathways. In addition, BBR markedly inhibited inflammation by reducing immune cell infiltration and inhibition of neutrophil activation and inflammatory gene expression. Furthermore, BBR was able to inhibit hepatic fibrosis by modulating the expression of multiple genes involved in hepatic stellate cell activation and cholangiocyte proliferation. Consistent with our previous findings, BBR's beneficial effects are linked with the downregulation of microRNA34a and long noncoding RNA H19, which are two important players in promoting NASH progression and liver fibrosis. Conclusion: BBR is a promising therapeutic agent for NASH by targeting multiple pathways. These results provide a strong foundation for a future clinical investigation.
Keywords: NAFLD; bile acids; fibrosis; inflammation.
Conflict of interest statement
The authors declare no competing financial interests.
Figures
Similar articles
-
Berberine alleviates nonalcoholic fatty liver induced by a high-fat diet in mice by activating SIRT3.FASEB J. 2019 Jun;33(6):7289-7300. doi: 10.1096/fj.201802316R. Epub 2019 Mar 8. FASEB J. 2019. PMID: 30848932
-
Berberine directly targets AKR1B10 protein to modulate lipid and glucose metabolism disorders in NAFLD.J Ethnopharmacol. 2024 Oct 5;332:118354. doi: 10.1016/j.jep.2024.118354. Epub 2024 May 17. J Ethnopharmacol. 2024. PMID: 38762210
-
Exploring the mechanism of berberine treatment for atherosclerosis combined with non-alcoholic fatty liver disease based on bioinformatic and experimental study.PLoS One. 2024 Dec 19;19(12):e0314961. doi: 10.1371/journal.pone.0314961. eCollection 2024. PLoS One. 2024. PMID: 39700090 Free PMC article.
-
The Potential Mechanisms of Berberine in the Treatment of Nonalcoholic Fatty Liver Disease.Molecules. 2016 Oct 14;21(10):1336. doi: 10.3390/molecules21101336. Molecules. 2016. PMID: 27754444 Free PMC article. Review.
-
Berberine prevents NAFLD and HCC by modulating metabolic disorders.Pharmacol Ther. 2024 Feb;254:108593. doi: 10.1016/j.pharmthera.2024.108593. Epub 2024 Feb 1. Pharmacol Ther. 2024. PMID: 38301771 Review.
Cited by
-
Yinzhihuang oral liquid protects against non-alcoholic steatohepatitis via modulation of the gut-liver axis in mice.Ann Transl Med. 2022 Jun;10(11):631. doi: 10.21037/atm-21-4809. Ann Transl Med. 2022. PMID: 35813333 Free PMC article.
-
Network Pharmacology and Bioinformatics Methods Reveal the Mechanism of Berberine in the Treatment of Ischaemic Stroke.Evid Based Complement Alternat Med. 2022 Jun 29;2022:5160329. doi: 10.1155/2022/5160329. eCollection 2022. Evid Based Complement Alternat Med. 2022. PMID: 35815278 Free PMC article.
-
Bile acid dysmetabolism in Bangladeshi infants is associated with poor linear growth, enteric inflammation, and small intestine bacterial overgrowth.medRxiv [Preprint]. 2025 Feb 5:2025.02.04.25321650. doi: 10.1101/2025.02.04.25321650. medRxiv. 2025. PMID: 39973996 Free PMC article. Preprint.
-
Gut Microbiome and Bile Acid Interactions: Mechanistic Implications for Cholangiocarcinoma Development, Immune Resistance, and Therapy.Am J Pathol. 2025 Mar;195(3):397-408. doi: 10.1016/j.ajpath.2024.11.004. Epub 2024 Dec 19. Am J Pathol. 2025. PMID: 39730075 Review.
-
Bile acid and receptors: biology and drug discovery for nonalcoholic fatty liver disease.Acta Pharmacol Sin. 2022 May;43(5):1103-1119. doi: 10.1038/s41401-022-00880-z. Epub 2022 Feb 25. Acta Pharmacol Sin. 2022. PMID: 35217817 Free PMC article. Review.
References
-
- Zhang Y.-P., Deng Y.-J., Tang K.-R., Chen R.-S., Liang S., Liang Y.-J., Han L., Jin L., Liang Z.-E., Chen Y.-N., et al. Berberine Ameliorates High-Fat Diet-Induced Non-Alcoholic Fatty Liver Disease in Rats via Activation of SIRT3/AMPK/ACC Pathway. Curr. Med. Sci. 2019;39:37–43. doi: 10.1007/s11596-019-1997-3. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

