Phylogenetic analysis of the caspase family in bivalves: implications for programmed cell death, immune response and development
- PMID: 33494703
- PMCID: PMC7836458
- DOI: 10.1186/s12864-021-07380-0
Phylogenetic analysis of the caspase family in bivalves: implications for programmed cell death, immune response and development
Abstract
Background: Apoptosis is an important process for an organism's innate immune system to respond to pathogens, while also allowing for cell differentiation and other essential life functions. Caspases are one of the key protease enzymes involved in the apoptotic process, however there is currently a very limited understanding of bivalve caspase diversity and function.
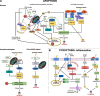
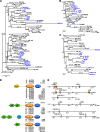
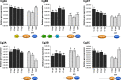
Results: In this work, we investigated the presence of caspase homologues using a combination of bioinformatics and phylogenetic analyses. We blasted the Crassostrea gigas genome for caspase homologues and identified 35 potential homologues in the addition to the already cloned 23 bivalve caspases. As such, we present information about the phylogenetic relationship of all identified bivalve caspases in relation to their homology to well-established vertebrate and invertebrate caspases. Our results reveal unexpected novelty and complexity in the bivalve caspase family. Notably, we were unable to identify direct homologues to the initiator caspase-9, a key-caspase in the vertebrate apoptotic pathway, inflammatory caspases (caspase-1, - 4 or - 5) or executioner caspases-3, - 6, - 7. We also explored the fact that bivalves appear to possess several unique homologues to the initiator caspase groups - 2 and - 8. Large expansions of caspase-3 like homologues (caspase-3A-C), caspase-3/7 group and caspase-3/7-like homologues were also identified, suggesting unusual roles of caspases with direct implications for our understanding of immune response in relation to common bivalve diseases. Furthermore, we assessed the gene expression of two initiator (Cg2A, Cg8B) and four executioner caspases (Cg3A, Cg3B, Cg3C, Cg3/7) in C. gigas late-larval development and during metamorphosis, indicating that caspase expression varies across the different developmental stages.
Conclusion: Our analysis provides the first overview of caspases across different bivalve species with essential new insights into caspase diversity, knowledge that can be used for further investigations into immune response to pathogens or regulation of developmental processes.
Keywords: Apoptosis; Bivalves; Caspase; Inflammation response; Innate immune system; Programmed cell death; Pyroptosis.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures

References
-
- Gerdol M, Gomez-Chiarri M, Castillo MG, Figueras A, Fiorito G, Moreira R, et al. Immunity in molluscs: recognition and effector mechanisms, with a focus on bivalvia. In: Cooper E, et al., editors. Advances in comparative immunology. Cham: Springer; 2018. pp. 225–341.
-
- Sokolova I. Apoptosis in molluscan immune defense. Invertebr Surviv J. 2009;6(1):49–58.
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

