ANO5 ensures trafficking of annexins in wounded myofibers
- PMID: 33496727
- PMCID: PMC7844426
- DOI: 10.1083/jcb.202007059
ANO5 ensures trafficking of annexins in wounded myofibers
Abstract
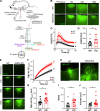
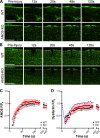
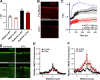
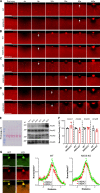
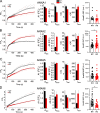
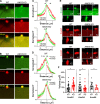
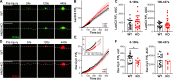
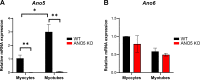
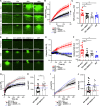
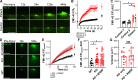
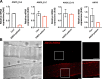
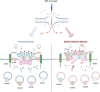
Mutations in ANO5 (TMEM16E) cause limb-girdle muscular dystrophy R12. Defective plasma membrane repair is a likely mechanism. Using myofibers from Ano5 knockout mice, we show that trafficking of several annexin proteins, which together form a cap at the site of injury, is altered upon loss of ANO5. Annexin A2 accumulates at the wound to nearly twice the level observed in WT fibers, while annexin A6 accumulation is substantially inhibited in the absence of ANO5. Appearance of annexins A1 and A5 at the cap is likewise diminished in the Ano5 knockout. These changes are correlated with an alteration in annexin repair cap fine structure and shedding of annexin-positive vesicles. We conclude that loss of annexin coordination during repair is disrupted in Ano5 knockout mice and underlies the defective repair phenotype. Although ANO5 is a phospholipid scramblase, abnormal repair is rescued by overexpression of a scramblase-defective ANO5 mutant, suggesting a novel, scramblase-independent role of ANO5 in repair.
© 2021 Foltz et al.
Figures
References
-
- Accardi, A. 2016. Mechanisms of Ion and Lipid Transport by TMEM16 Scramblases. Biophys. J. 110:174a. 10.1016/j.bpj.2015.11.968 - DOI
-
- Andreeva, T.V., Tyazhelova T.V., Rykalina V.N., Gusev F.E., Goltsov A.Y., Zolotareva O.I., Aliseichik M.P., Borodina T.A., Grigorenko A.P., Reshetov D.A., et al. . 2016. Whole exome sequencing links dental tumor to an autosomal-dominant mutation in ANO5 gene associated with gnathodiaphyseal dysplasia and muscle dystrophies. Sci. Rep. 6:26440. 10.1038/srep26440 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

