Adjuvant Selection for Influenza and RSV Prefusion Subunit Vaccines
- PMID: 33498370
- PMCID: PMC7909420
- DOI: 10.3390/vaccines9020071
Adjuvant Selection for Influenza and RSV Prefusion Subunit Vaccines
Abstract
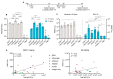
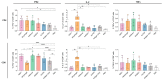
Subunit vaccines exhibit favorable safety and immunogenicity profiles and can be designed to mimic native antigen structures. However, pairing with an appropriate adjuvant is imperative in order to elicit effective humoral and cellular immune responses. In this study, we aimed to determine an optimal adjuvant pairing with the prefusion form of influenza haemagglutinin (HA) or respiratory syncytial virus (RSV) fusion (F) subunit vaccines in BALB/c mice in order to inform future subunit vaccine adjuvant selection. We tested a panel of adjuvants, including aluminum hydroxide (alhydrogel), QS21, Addavax, Addavax with QS21 (AdQS21), and Army Liposome Formulation 55 with monophosphoryl lipid A and QS21 (ALF55). We found that all adjuvants elicited robust humoral responses in comparison to placebo, with the induction of potent neutralizing antibodies observed in all adjuvanted groups against influenza and in AdQS21, alhydrogel, and ALF55 against RSV. Upon HA vaccination, we observed that none of the adjuvants were able to significantly increase the frequency of CD4+ and CD8+ IFN-γ+ cells when compared to unadjuvanted antigen. The varying responses to antigens with each adjuvant highlights that those adjuvants most suited for pairing purposes can vary depending on the antigen used and/or the desired immune response. We therefore suggest that an adjuvant trial for different subunit vaccines in development would likely be necessary in preclinical studies.
Keywords: RSV; adjuvant; influenza; microbiology; vaccine; virology.
Conflict of interest statement
K.J.C., D.W., and P.R.Y. are inventors of the “Molecular Clamp” patent, US 2020/0040042.
Figures

Similar articles
-
Modified mRNA/lipid nanoparticle-based vaccines expressing respiratory syncytial virus F protein variants are immunogenic and protective in rodent models of RSV infection.NPJ Vaccines. 2020 Feb 14;5(1):16. doi: 10.1038/s41541-020-0163-z. eCollection 2020. NPJ Vaccines. 2020. PMID: 32128257 Free PMC article.
-
A novel respiratory syncytial virus (RSV) F subunit vaccine adjuvanted with GLA-SE elicits robust protective TH1-type humoral and cellular immunity in rodent models.PLoS One. 2015 Mar 20;10(3):e0119509. doi: 10.1371/journal.pone.0119509. eCollection 2015. PLoS One. 2015. PMID: 25793508 Free PMC article.
-
Squalene-adjuvanted H7N9 virus vaccine induces robust humoral immune response against H7N9 and H7N7 viruses.Vaccine. 2014 Jul 31;32(35):4485-4494. doi: 10.1016/j.vaccine.2014.06.043. Epub 2014 Jun 21. Vaccine. 2014. PMID: 24962757
-
Army Liposome Formulation (ALF) family of vaccine adjuvants.Expert Rev Vaccines. 2020 Mar;19(3):279-292. doi: 10.1080/14760584.2020.1745636. Epub 2020 Mar 31. Expert Rev Vaccines. 2020. PMID: 32228108 Free PMC article. Review.
-
Adjuvants are Key Factors for the Development of Future Vaccines: Lessons from the Finlay Adjuvant Platform.Front Immunol. 2013 Dec 2;4:407. doi: 10.3389/fimmu.2013.00407. Front Immunol. 2013. PMID: 24348475 Free PMC article. Review.
Cited by
-
Vaccine and therapeutic agents against the respiratory syncytial virus: resolved and unresolved issue.MedComm (2020). 2024 Nov 21;5(12):e70016. doi: 10.1002/mco2.70016. eCollection 2024 Dec. MedComm (2020). 2024. PMID: 39575302 Free PMC article. Review.
-
Natural killer cells contribute to enhanced respiratory disease after oil-in-water emulsion adjuvanted vaccination against respiratory syncytial virus and infection.Hum Vaccin Immunother. 2021 Oct 3;17(10):3806-3817. doi: 10.1080/21645515.2021.1915039. Epub 2021 Apr 20. Hum Vaccin Immunother. 2021. PMID: 33877948 Free PMC article.
-
LDH as an adjuvant makes Brucella outer-membrane vesicles and outer-membrane vesicle-associated proteins highly protective in mice.Iran J Basic Med Sci. 2023;26(5):564-571. doi: 10.22038/IJBMS.2023.67394.14775. Iran J Basic Med Sci. 2023. PMID: 37051103 Free PMC article.
-
Development of molecular clamp stabilized hemagglutinin vaccines for Influenza A viruses.NPJ Vaccines. 2021 Nov 8;6(1):135. doi: 10.1038/s41541-021-00395-4. NPJ Vaccines. 2021. PMID: 34750396 Free PMC article.
-
Immunogenicity of an AS01-adjuvanted respiratory syncytial virus prefusion F (RSVPreF3) vaccine in animal models.NPJ Vaccines. 2023 Sep 29;8(1):143. doi: 10.1038/s41541-023-00729-4. NPJ Vaccines. 2023. PMID: 37773185 Free PMC article.
References
-
- Knudsen N.P.H., Olsen A., Buonsanti C., Follmann F., Zhang Y., Coler R.N., Fox C.B., Meinke A., D´Oro U., Casini D., et al. Different human vaccine adjuvants promote distinct antigen-independent immunological signatures tailored to different pathogens. Sci. Rep. 2016;6:19570. doi: 10.1038/srep19570. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

