Function-Oriented and Modular (+/-)-cis-Pseudoguaianolide Synthesis: Discovery of New Nrf2 Activators and NF-κB Inhibitors
- PMID: 33502811
- PMCID: PMC8195264
- DOI: 10.1002/chem.202100038
Function-Oriented and Modular (+/-)-cis-Pseudoguaianolide Synthesis: Discovery of New Nrf2 Activators and NF-κB Inhibitors
Abstract
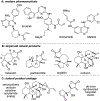
Described herein is a function-oriented synthesis route and biological evaluation of pseudoguaianolide analogues. The 10-step synthetic route developed retains the topological complexity of the natural product, installs functional handles for late-stage diversification, and forges the key bioactive Michael acceptors early in the synthesis. The analogues were found to be low-micromolar Nrf2 activators and micromolar NF-κB inhibitors and dependent on the local environment of the Michael acceptor moieties.
Keywords: NF-κB inhibitors; Nrf2 activators; enyne metathesis; modular synthesis; pseudoguaianolides.
© 2021 Wiley-VCH GmbH.
Conflict of interest statement
Conflict of interest
The authors declare no conflict of interest.
Figures

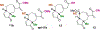
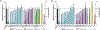






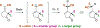

References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

