Contributions of Flow Cytometry to the Molecular Study of Spermatogenesis in Mammals
- PMID: 33503798
- PMCID: PMC7865295
- DOI: 10.3390/ijms22031151
Contributions of Flow Cytometry to the Molecular Study of Spermatogenesis in Mammals
Abstract
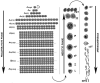
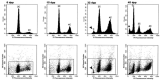
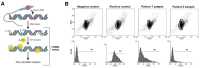
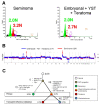
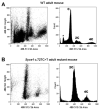
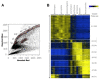
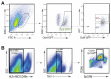
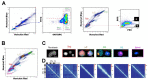
Mammalian testes are very heterogeneous organs, with a high number of different cell types. Testicular heterogeneity, together with the lack of reliable in vitro culture systems of spermatogenic cells, have been an obstacle for the characterization of the molecular bases of the unique events that take place along the different spermatogenic stages. In this context, flow cytometry has become an invaluable tool for the analysis of testicular heterogeneity, and for the purification of stage-specific spermatogenic cell populations, both for basic research and for clinical applications. In this review, we highlight the importance of flow cytometry for the advances on the knowledge of the molecular groundwork of spermatogenesis in mammals. Moreover, we provide examples of different approaches to the study of spermatogenesis that have benefited from flow cytometry, including the characterization of mutant phenotypes, transcriptomics, epigenetic and genome-wide chromatin studies, and the attempts to establish cell culture systems for research and/or clinical aims such as infertility treatment.
Keywords: FACS; flow cytometry; male infertility; spermatogenesis.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

