Analysis of a Preliminary microRNA Expression Signature in a Human Telangiectatic Osteogenic Sarcoma Cancer Cell Line
- PMID: 33503899
- PMCID: PMC7866083
- DOI: 10.3390/ijms22031163
Analysis of a Preliminary microRNA Expression Signature in a Human Telangiectatic Osteogenic Sarcoma Cancer Cell Line
Abstract
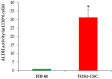
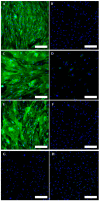
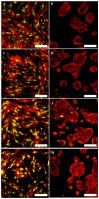
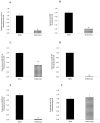
Telangiectatic osteosarcoma (TOS) is an aggressive variant of osteosarcoma (OS) with distinctive radiographic, gross, microscopic features, and prognostic implications. Despite several studies on OS, we are still far from understanding the molecular mechanisms of TOS. In recent years, many studies have demonstrated not only that microRNAs (miRNAs) are involved in OS tumorigenesis, development, and metastasis, but also that the presence in high-grade types of OS of cancer stem cells (CSCs) plays an important role in tumor progression. Despite these findings, nothing has been described previously about the expression of miRNAs and the presence of CSCs in human TOS. Therefore, we have isolated/characterized a putative CSC cell line from human TOS (TOS-CSCs) and evaluated the expression levels of several miRNAs in TOS-CSCs using real-time quantitative assays. We show, for the first time, the existence of CSCs in human TOS, highlighting the in vitro establishment of this unique stabilized cell line and an identification of a preliminary expression of the miRNA profile, characteristic of TOS-CSCs. These findings represent an important step in the study of the biology of one of the most aggressive variants of OS and the role of miRNAs in TOS-CSC behavior.
Keywords: cancer stem cells; microRNAs; telangiectatic osteosarcoma.
Conflict of interest statement
The authors declare no conflict of interest.
Figures









Similar articles
-
Genetic and molecular characterization of the human osteosarcoma 3AB-OS cancer stem cell line: a possible model for studying osteosarcoma origin and stemness.J Cell Physiol. 2013 Jun;228(6):1189-201. doi: 10.1002/jcp.24272. J Cell Physiol. 2013. PMID: 23129384
-
MiR-9 is overexpressed in spontaneous canine osteosarcoma and promotes a metastatic phenotype including invasion and migration in osteoblasts and osteosarcoma cell lines.BMC Cancer. 2016 Oct 10;16(1):784. doi: 10.1186/s12885-016-2837-5. BMC Cancer. 2016. PMID: 27724924 Free PMC article.
-
MicroRNA-29b-1 impairs in vitro cell proliferation, self‑renewal and chemoresistance of human osteosarcoma 3AB-OS cancer stem cells.Int J Oncol. 2014 Nov;45(5):2013-23. doi: 10.3892/ijo.2014.2618. Epub 2014 Aug 22. Int J Oncol. 2014. PMID: 25174983 Free PMC article.
-
MicroRNAs for osteosarcoma in the mouse: a meta-analysis.Oncotarget. 2016 Dec 20;7(51):85650-85674. doi: 10.18632/oncotarget.13333. Oncotarget. 2016. PMID: 27852052 Free PMC article. Review.
-
Autophagy regulation by microRNAs: Novel insights into osteosarcoma therapy.IUBMB Life. 2020 Jul;72(7):1306-1321. doi: 10.1002/iub.2277. Epub 2020 Mar 31. IUBMB Life. 2020. PMID: 32233112 Review.
Cited by
-
Cancer Stem Cells in Sarcomas: In Vitro Isolation and Role as Prognostic Markers: A Systematic Review.Cancers (Basel). 2023 Apr 25;15(9):2449. doi: 10.3390/cancers15092449. Cancers (Basel). 2023. PMID: 37173919 Free PMC article. Review.
-
Feasibility and barriers to rapid establishment of patient-derived primary osteosarcoma cell lines in clinical management.iScience. 2024 Jun 19;27(9):110251. doi: 10.1016/j.isci.2024.110251. eCollection 2024 Sep 20. iScience. 2024. PMID: 39286504 Free PMC article. Review.
-
Establishment and Molecular Characterization of a Human Stem Cell Line from a Primary Cell Culture Obtained from an Ectopic Calcified Lesion of a Tumoral Calcinosis Patient Carrying a Novel GALNT3 Mutation.Genes (Basel). 2025 Feb 24;16(3):263. doi: 10.3390/genes16030263. Genes (Basel). 2025. PMID: 40149415 Free PMC article.
References
-
- Bielack S.S., Kempf-Bielack B., Delling G., Exner G.U., Flege S., Helmke K., Kotz R., Salzer-Kuntschik M., Werner M., Winkelmann W., et al. Prognostic factors in high grade osteosarcoma of the extremities or trunk: An analysis of 1702 patients treated on neoadjuvant cooperative osteosarcoma study group protocols. J. Clin. Oncol. 2002;20:776–790. doi: 10.1200/JCO.2002.20.3.776. - DOI - PubMed
-
- Geller D.S., Gorlik R. Osteosarcoma: A review of diagnosis, management, and treatment strategies. Clin. Adv. Haematol. Oncol. 2010;8:705–718. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

