Filgotinib versus placebo or adalimumab in patients with rheumatoid arthritis and inadequate response to methotrexate: a phase III randomised clinical trial
- PMID: 33504485
- PMCID: PMC8237199
- DOI: 10.1136/annrheumdis-2020-219214
Filgotinib versus placebo or adalimumab in patients with rheumatoid arthritis and inadequate response to methotrexate: a phase III randomised clinical trial
Abstract
Objective: To evaluate the efficacy and safety of the Janus kinase-1-preferential inhibitor filgotinib versus placebo or tumour necrosis factor-α inhibitor therapy in patients with active rheumatoid arthritis (RA) despite ongoing treatment with methotrexate (MTX).
Methods: This 52-week, multicentre, double-blind, placebo-controlled and active-controlled phase III trial evaluated once-daily oral filgotinib in patients with RA randomised 3:3:2:3 to filgotinib 200 mg (FIL200) or filgotinib 100 mg (FIL100), subcutaneous adalimumab 40 mg biweekly, or placebo (through week 24), all with stable weekly background MTX. The primary endpoint was the proportion of patients achieving 20% improvement in American College of Rheumatology criteria (ACR20) at week 12. Additional efficacy outcomes were assessed sequentially. Safety was assessed from adverse events and laboratory abnormalities.
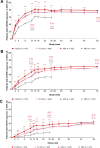
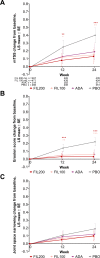
Results: The proportion of patients (n=1755 randomised and treated) achieving ACR20 at week 12 was significantly higher for FIL200 (76.6%) and FIL100 (69.8%) versus placebo (49.9%; treatment difference (95% CI), 26.7% (20.6% to 32.8%) and 19.9% (13.6% to 26.2%), respectively; both p<0.001). Filgotinib was superior to placebo in key secondary endpoints assessing RA signs and symptoms, physical function and structural damage. FIL200 was non-inferior to adalimumab in terms of Disease Activity Score in 28 joints with C reactive protein ≤3.2 at week 12 (p<0.001); FIL100 did not achieve non-inferiority. Adverse events and laboratory abnormalities were comparable among active treatment arms.
Conclusions: Filgotinib improved RA signs and symptoms, improved physical function, inhibited radiographic progression and was well tolerated in patients with RA with inadequate response to MTX. FIL200 was non-inferior to adalimumab.
Trial registration number: NCT02889796.
Keywords: antirheumatic agents; arthritis; rheumatoid; therapeutics.
© Author(s) (or their employer(s)) 2021. Re-use permitted under CC BY-NC. No commercial re-use. See rights and permissions. Published by BMJ.
Conflict of interest statement
Competing interests: BC received honoraria from AbbVie, Bristol-Myers Squibb, Gilead Sciences, Janssen, Lilly, Merck, Novartis, Pfizer, Roche-Chugai, Sanofi and UCB; and research grants from Novartis, Pfizer and Roche. AK received honoraria or consulting fees from AbbVie, Boehringer Ingelheim, Flexion, Genzyme, Gilead Sciences, Janssen, Novartis, Pfizer, Regeneron, Sanofi and Sun Pharma Advanced Research; was a paid instructor or speaker for AbbVie, Celgene, Flexion, Genzyme, Horizon, Merck, Novartis, Pfizer, Regeneron and Sanofi; and holds shares in Amgen, Gilead Sciences, GlaxoSmithKline, Pfizer and Sanofi. YT has received speaking fees and/or honoraria from AbbVie, Asahi Kasei, Astellas, Bristol-Myers, Chugai, Daiichi Sankyo, Eisai, Eli Lilly, Gilead, GSK, Janssen, Mitsubishi Tanabe, Novartis, Pfizer, Sanofi and YL Biologics; and research grants from AbbVie, Chugai, Daiichi Sankyo, Eisai, Mitsubishi Tanabe, Takeda and UCB. DvdH received consulting fees from AbbVie, Amgen, Astellas, AstraZeneca, BMS, Boehringer Ingelheim, Celgene, Cyxone, Daiichi Sankyo, Eisai, Eli Lilly, Galapagos, Gilead, GlaxoSmithKline, Janssen, Merck, Novartis, Pfizer, Regeneron, Roche, Sanofi, Takeda and UCB Pharma; and is the Director of Imaging Rheumatology BV. JAS, UK and S-CB report nothing to disclose. HSBB has received honoraria or consulting fees from AbbVie, Gilead Sciences, Horizon and Merck; and research grants or support from AbbVie, Sanofi, Regeneron, Eli Lilly, Pfizer, Selecta Biosciences, Gilead Sciences, Horizon, Janssen and Pfizer. FM, BB, LY and YG are employees and shareholders of Gilead Sciences. MCG has received honoraria or consulting fees from AbbVie, Amgen, BeiGene, Genentech, Gilead Sciences, Lilly Pharmaceuticals, Sanofi Genzyme, RPharm and SetPoint. He is also an employee and shareholder of Gilead Sciences. CT is an employee and shareholder of Galapagos NV. JSS, AJ and NM are former employees of Gilead Sciences and may hold shares. RBML has received honoraria or consulting fees from AbbVie, AstraZeneca, Bristol-Myers Squibb, Eli Lilly, Galapagos NV, Novartis, Pfizer and UCB. EK has received honoraria or consulting fees from AbbVie, Amgen, AstraZeneca, Bristol-Myers Squibb, Celltrion, F Hoffmann-La Roche, Genentech, Gilead Sciences, Janssen, Lilly Pharmaceuticals, Merck, Myriad Autoimmune, Pfizer, Sandoz, Sanofi Genzyme and Samsung Bioepis; has received speaking fees from AbbVie, Amgen, Bristol-Myers Squibb, F Hoffmann-La Roche, Janssen, Merck, Pfizer, Sanofi Genzyme and UCB; and has received research grants or support from AbbVie, Amgen, Gilead Sciences, Lilly Pharmaceuticals, Merck, Pfizer, PuraPharm and Sanofi. PN has received honoraria or consulting fees, grants or research support, or been a member of a speakers bureau for AbbVie, Bristol-Myers Squibb, Celgene, Gilead Sciences, Janssen, Lilly, MSD, Novartis, Pfizer, Roche, Sanofi and UCB.
Figures


References
-
- Furst DE, Pangan AL, Harrold LR, et al. Greater likelihood of remission in rheumatoid arthritis patients treated earlier in the disease course: results from the Consortium of rheumatology researchers of North America registry. Arthritis Care Res 2011;63:856–64. 10.1002/acr.20452 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

