Next-Generation Immunotherapies to Improve Anticancer Immunity
- PMID: 33505304
- PMCID: PMC7831045
- DOI: 10.3389/fphar.2020.566401
Next-Generation Immunotherapies to Improve Anticancer Immunity
Abstract
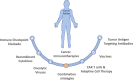
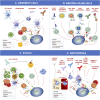
Checkpoint inhibitors are widely used immunotherapies for advanced cancer. Nonetheless, checkpoint inhibitors have a relatively low response rate, work in a limited range of cancers, and have some unignorable side effects. Checkpoint inhibitors aim to reinvigorate exhausted or suppressed T cells in the tumor microenvironment (TME). However, the TME contains various other immune cell subsets that interact to determine the fate of cytotoxic T cells. Activation of cytotoxic T cells is initiated by antigen cross-presentation of dendritic cells. Dendritic cells could also release chemokines and cytokines to recruit and foster T cells. B cells, another type of antigen-presenting cell, also foster T cells and can produce tumor-specific antibodies. Neutrophils, a granulocyte cell subset in the TME, impede the proliferation and activation of T cells. The TME also consists of cytotoxic innate natural killer cells, which kill tumor cells efficiently. Natural killer cells can eradicate major histocompatibility complex I-negative tumor cells, which escape cytotoxic T cell-mediated destruction. A thorough understanding of the immune mechanism of the TME, as reviewed here, will lead to further development of more powerful therapeutic strategies. We have also reviewed the clinical outcomes of patients treated with drugs targeting these immune cells to identify strategies for improvement and possible immunotherapy combinations.
Keywords: B cells; DC; NK cells; combination immunotherapy; neutrophils.
Copyright © 2021 Shi, Tomczak, Li, Ochieng, Lee and Haymaker.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Similar articles
-
Cytokines Orchestrating the Natural Killer-Myeloid Cell Crosstalk in the Tumor Microenvironment: Implications for Natural Killer Cell-Based Cancer Immunotherapy.Front Immunol. 2021 Jan 29;11:621225. doi: 10.3389/fimmu.2020.621225. eCollection 2020. Front Immunol. 2021. PMID: 33584718 Free PMC article. Review.
-
Targeting Checkpoint Receptors and Molecules for Therapeutic Modulation of Natural Killer Cells.Front Immunol. 2018 Sep 10;9:2041. doi: 10.3389/fimmu.2018.02041. eCollection 2018. Front Immunol. 2018. PMID: 30250471 Free PMC article. Review.
-
Harnessing the cDC1-NK Cross-Talk in the Tumor Microenvironment to Battle Cancer.Front Immunol. 2021 Feb 19;11:631713. doi: 10.3389/fimmu.2020.631713. eCollection 2020. Front Immunol. 2021. PMID: 33679726 Free PMC article. Review.
-
CD8+ cytotoxic T lymphocytes in cancer immunotherapy: A review.J Cell Physiol. 2019 Jun;234(6):8509-8521. doi: 10.1002/jcp.27782. Epub 2018 Nov 22. J Cell Physiol. 2019. PMID: 30520029 Review.
-
Immune and Inflammatory Cells in Thyroid Cancer Microenvironment.Int J Mol Sci. 2019 Sep 7;20(18):4413. doi: 10.3390/ijms20184413. Int J Mol Sci. 2019. PMID: 31500315 Free PMC article. Review.
Cited by
-
Modulating gut microbiome in cancer immunotherapy: Harnessing microbes to enhance treatment efficacy.Cell Rep Med. 2024 Apr 16;5(4):101478. doi: 10.1016/j.xcrm.2024.101478. Cell Rep Med. 2024. PMID: 38631285 Free PMC article. Review.
-
Regulation of tumor immunity and immunotherapy by the tumor collagen extracellular matrix.Front Immunol. 2023 Aug 17;14:1199513. doi: 10.3389/fimmu.2023.1199513. eCollection 2023. Front Immunol. 2023. PMID: 37662958 Free PMC article. Review.
-
Metabolic Modifications, Inflammation, and Cancer Immunotherapy.Front Oncol. 2021 Sep 24;11:703681. doi: 10.3389/fonc.2021.703681. eCollection 2021. Front Oncol. 2021. PMID: 34631531 Free PMC article. Review.
-
Next-Generation Immunotherapy: Advancing Clinical Applications in Cancer Treatment.J Clin Med. 2024 Oct 30;13(21):6537. doi: 10.3390/jcm13216537. J Clin Med. 2024. PMID: 39518676 Free PMC article.
-
RAAS: A Convergent Player in Ischemic Heart Failure and Cancer.Int J Mol Sci. 2021 Jul 1;22(13):7106. doi: 10.3390/ijms22137106. Int J Mol Sci. 2021. PMID: 34281199 Free PMC article. Review.
References
-
- Agnello G., Alters S. E., Lowe D. G., Rowlinson S. W. (2017). Abstract 3964: reducing systemic arginine with arginase (AEB1102) therapy does not suppress the immune response induced by anti-PD-1 and anti-PD-L1, and exerts an additive anti-tumor and synergistic survival benefit. Cancer Res. 77, 3964.
-
- Ajona D., Ortiz-Espinosa S., Moreno H., Lozano T., Pajares M. J., Agorreta J., et al. (2017). A combined PD-1/C5a blockade synergistically protects against lung cancer growth and metastasis. Cancer Discov. 7, 694–703. 10.1158/2159-8290.cd-16-1184 - DOI - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

