Velvet Antler Methanol Extracts Ameliorate Parkinson's Disease by Inhibiting Oxidative Stress and Neuroinflammation: From C. elegans to Mice
- PMID: 33505591
- PMCID: PMC7811427
- DOI: 10.1155/2021/8864395
Velvet Antler Methanol Extracts Ameliorate Parkinson's Disease by Inhibiting Oxidative Stress and Neuroinflammation: From C. elegans to Mice
Abstract
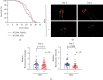
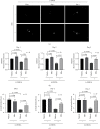
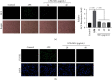
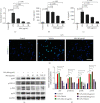
Velvet antler is the traditional tonic food or medicine used in East Asia for treating aging-related diseases. Herein, we try to dissect the pharmacology of methanol extracts (MEs) of velvet antler on Parkinson's disease (PD). Caenorhabditis elegans studies showed that MEs decreased the aggregation of α-synuclein and protected oxidative stress-induced DAergic neuron degeneration. In vitro cellular data indicated that MEs suppressed the LPS-induced MAPKs and NF-κB activation, therefore inhibiting overproduction of reactive oxygen species, nitric oxide, tumor necrosis factor-α, and interleukin-6; blocking microglia activation; and protecting DAergic neurons from the microglia-mediated neurotoxicity. In vivo MPTP-induced PD mouse investigations found that MEs prevented MPTP-induced neuron loss in the substantia nigra and improved the behavioral rotating rod performance in MPTP-treated mice by increasing the expression level of tyrosine hydroxylase (TH) and downregulating α-synuclein protein expression. In all, these results demonstrate that MEs ameliorate PD by inhibiting oxidative stress and neuroinflammation.
Copyright © 2021 Ying Liu et al.
Conflict of interest statement
The authors declare that they have no conflicts of interest.
Figures


Similar articles
-
Velvet antler methanol extracts (MEs) protects against oxidative stress in Caenorhabditis elegans by SKN-1.Biomed Pharmacother. 2020 Jan;121:109668. doi: 10.1016/j.biopha.2019.109668. Epub 2019 Nov 19. Biomed Pharmacother. 2020. PMID: 31766103
-
5-(3,4-Difluorophenyl)-3-(6-methylpyridin-3-yl)-1,2,4-oxadiazole (DDO-7263), a novel Nrf2 activator targeting brain tissue, protects against MPTP-induced subacute Parkinson's disease in mice by inhibiting the NLRP3 inflammasome and protects PC12 cells against oxidative stress.Free Radic Biol Med. 2019 Apr;134:288-303. doi: 10.1016/j.freeradbiomed.2019.01.003. Epub 2019 Jan 5. Free Radic Biol Med. 2019. PMID: 30615919
-
Neuroprotective effects of Astilbin on MPTP-induced Parkinson's disease mice: Glial reaction, α-synuclein expression and oxidative stress.Int Immunopharmacol. 2019 Jan;66:19-27. doi: 10.1016/j.intimp.2018.11.004. Epub 2018 Nov 9. Int Immunopharmacol. 2019. PMID: 30419450
-
Pathological α-synuclein exacerbates the progression of Parkinson's disease through microglial activation.Toxicol Lett. 2017 Jan 4;265:30-37. doi: 10.1016/j.toxlet.2016.11.002. Epub 2016 Nov 16. Toxicol Lett. 2017. PMID: 27865851 Review.
-
Reprint of: revisiting oxidative stress and mitochondrial dysfunction in the pathogenesis of Parkinson disease-resemblance to the effect of amphetamine drugs of abuse.Free Radic Biol Med. 2013 Sep;62:186-201. doi: 10.1016/j.freeradbiomed.2013.05.042. Epub 2013 Jun 3. Free Radic Biol Med. 2013. PMID: 23743292 Review.
Cited by
-
Sika Deer Velvet Antler Peptide Exerts Neuroprotective Effect in a Parkinson's Disease Model via Regulating Oxidative Damage and Gut Microbiota.Pharmaceuticals (Basel). 2024 Jul 22;17(7):972. doi: 10.3390/ph17070972. Pharmaceuticals (Basel). 2024. PMID: 39065820 Free PMC article.
-
Studying the mechanisms of neurodegeneration: C. elegans advantages and opportunities.Front Cell Neurosci. 2025 Mar 26;19:1559151. doi: 10.3389/fncel.2025.1559151. eCollection 2025. Front Cell Neurosci. 2025. PMID: 40207239 Free PMC article. Review.
-
SIRT3 expression alleviates microglia activation‑induced dopaminergic neuron injury through the mitochondrial pathway.Exp Ther Med. 2022 Sep 7;24(5):662. doi: 10.3892/etm.2022.11598. eCollection 2022 Nov. Exp Ther Med. 2022. PMID: 36168411 Free PMC article.
-
Velvet antler water extract protects porcine oocytes from lipopolysaccharide-induced meiotic defects.Cell Prolif. 2023 Jul;56(7):e13392. doi: 10.1111/cpr.13392. Epub 2023 Jan 3. Cell Prolif. 2023. PMID: 36596647 Free PMC article.
-
Docosahexaenoic Acid (DHA) Supplementation in a Triglyceride Form Prevents from Polyglutamine-Induced Dysfunctions in Caenorhabditis elegans.Int J Mol Sci. 2024 Nov 23;25(23):12594. doi: 10.3390/ijms252312594. Int J Mol Sci. 2024. PMID: 39684306 Free PMC article.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

