Comparative genomics of cetartiodactyla: energy metabolism underpins the transition to an aquatic lifestyle
- PMID: 33505701
- PMCID: PMC7816800
- DOI: 10.1093/conphys/coaa136
Comparative genomics of cetartiodactyla: energy metabolism underpins the transition to an aquatic lifestyle
Abstract
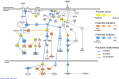
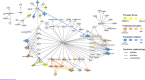
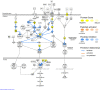
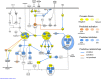
Foraging disruption caused by human activities is emerging as a key issue in cetacean conservation because it can affect nutrient levels and the amount of energy available to individuals to invest into reproduction. Our ability to predict how anthropogenic stressors affect these ecological processes and ultimately population trajectory depends crucially on our understanding of the complex physiological mechanisms that detect nutrient availability and regulate energy metabolism, foraging behavior and life-history decisions. These physiological mechanisms are likely to differ considerably from terrestrial mammalian model systems. Here, we examine nucleotide substitution rates in cetacean and other artiodactyl genomes to identify signatures of selection in genes associated with nutrient sensing pathways. We also estimated the likely physiological consequences of adaptive amino acid substitutions for pathway functions. Our results highlight that genes involved in the insulin, mTOR and NF-ĸB pathways are subject to significant positive selection in cetaceans compared to terrestrial artiodactyla. These genes may have been positively selected to enable cetaceans to adapt to a glucose-poor diet, to overcome deleterious effects caused by hypoxia during diving (e.g. oxidative stress and inflammation) and to modify fat-depot signaling functions in a manner different to terrestrial mammals. We thus show that adaptation in cetaceans to an aquatic lifestyle significantly affected functions in nutrient sensing pathways. The use of fat stores as a condition index in cetaceans may be confounded by the multiple and critical roles fat has in regulating cetacean metabolism, foraging behavior and diving physiology.
Keywords: Cetaceans; NF-ĸB; energy metabolism; evolution; insulin; mTOR.
© The Author(s) 2021. Published by Oxford University Press and the Society for Experimental Biology.
Figures

References
-
- Arkan MC, Hevener AL, Greten FR, Maeda S, Li Z-W, Long JM, Wynshaw-Boris A, Poli G, Olefsky J, Karin M (2005) IKK-beta links inflammation to obesity-induced insulin resistance. Nat Med 11: 191–198. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

