Increased Circulating of CD54highCD181low Neutrophils in Myelodysplastic Syndrome
- PMID: 33505909
- PMCID: PMC7830137
- DOI: 10.3389/fonc.2020.585216
Increased Circulating of CD54highCD181low Neutrophils in Myelodysplastic Syndrome
Abstract
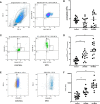
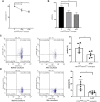
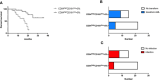
Myelodysplastic syndromes (MDSs) are a group of heterogeneous hematopoietic stem/progenitor cells clonal diseases, characteristic features with myeloid dysplasia, leading to abnormality of neutrophils. Recent studied have showed that neutrophils act not only as professional killers, but also as regulators of innate and adaptive immune in infection and inflammatory condition. The CD54highCD181low neutrophils are a kind of reverse-transmigrated neutrophils characterized proinflammatory phenotype. We investigated the frequency and functional properties of circulating CD54highCD181low neutrophils in patients with untreated MDS. Frequency of CD54highCD181low neutrophils was significantly increased in MDS patients and related to the severity of the disease. Furthermore, CD54highCD181low neutrophils suppressed CD8+ T cells functions in vitro. CD54highCD181low neutrophils lead to upregulation of PD1 on CD8+ T cells. Higher CD54highCD181low neutrophils were related to poor prognosis and more infections. The frequency of CD54highCD181low neutrophils decreased in high risk MDS patients who had response after treatment with decitabine. Overall, we identified CD54highCD181low neutrophils expanded in MDS. The exact mechanisms of increased CD54highCD181low neutrophils and its effect on immune function remain to be elucidated.
Keywords: CD181; CD54/ICAM-1; immunity; myelodysplastic syndromes; neutrophils.
Copyright © 2021 Yang, Li, Liu, Xie, Zhang, Niu, Shao, Xing and Wang.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


Similar articles
-
Distinct Neutrophil Populations in the Spleen During PICS.Front Immunol. 2020 May 15;11:804. doi: 10.3389/fimmu.2020.00804. eCollection 2020. Front Immunol. 2020. PMID: 32499777 Free PMC article.
-
Physician Education: Myelodysplastic Syndrome.Oncologist. 1996;1(4):284-287. Oncologist. 1996. PMID: 10388004
-
Deregulation of innate immune and inflammatory signaling in myelodysplastic syndromes.Leukemia. 2015 Jul;29(7):1458-69. doi: 10.1038/leu.2015.69. Epub 2015 Mar 12. Leukemia. 2015. PMID: 25761935 Free PMC article. Review.
-
A Study of Human Leukocyte Antigen Mismatched Cellular Therapy (Stem Cell Microtransplantation) in High-Risk Myelodysplastic Syndrome or Transformed Acute Myelogenous Leukemia.Stem Cells Transl Med. 2016 Apr;5(4):524-9. doi: 10.5966/sctm.2015-0196. Epub 2016 Feb 2. Stem Cells Transl Med. 2016. PMID: 26838271 Free PMC article. Clinical Trial.
-
Dendritic cells in myelodysplastic syndromes: from pathogenesis to immunotherapy.Immunotherapy. 2013 Jun;5(6):621-37. doi: 10.2217/imt.13.51. Immunotherapy. 2013. PMID: 23725285 Review.
Cited by
-
Immune-monitoring of myelodysplastic neoplasms: Recommendations from the i4MDS consortium.Hemasphere. 2024 May 15;8(5):e64. doi: 10.1002/hem3.64. eCollection 2024 May. Hemasphere. 2024. PMID: 38756352 Free PMC article.
-
Neutropenia (even mild) and anemia are poor prognostic factors in myelodysplastic syndromes.Front Med (Lausanne). 2025 Jul 11;12:1558585. doi: 10.3389/fmed.2025.1558585. eCollection 2025. Front Med (Lausanne). 2025. PMID: 40718416 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

