Biomimetic Tympanic Membrane Replacement Made by Melt Electrowriting
- PMID: 33506636
- PMCID: PMC11468533
- DOI: 10.1002/adhm.202002089
Biomimetic Tympanic Membrane Replacement Made by Melt Electrowriting
Abstract
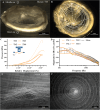
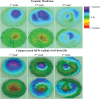
The tympanic membrane (TM) transfers sound waves from the air into mechanical motion for the ossicular chain. This requires a high sensitivity to small dynamic pressure changes and resistance to large quasi-static pressure differences. The TM achieves this by providing a layered structure of about 100µm in thickness, a low flexural stiffness, and a high tensile strength. Chronically infected middle ears require reconstruction of a large area of the TM. However, current clinical treatment can cause a reduction in hearing. With the novel additive manufacturing technique of melt electrowriting (MEW), it is for the first time possible to fabricate highly organized and biodegradable membranes within the dimensions of the TM. Scaffold designs of various fiber composition are analyzed mechanically and acoustically. It can be demonstrated that by customizing fiber orientation, fiber diameter, and number of layers the desired properties of the TM can be met. An applied thin collagen layer seals the micropores of the MEW-printed membrane while keeping the favorable mechanical and acoustical characteristics. The determined properties are beneficial for implantation, closely match those of the human TM, and support the growth of a neo-epithelial layer. This proves the possibilities to create a biomimimetic TM replacement using MEW.
Keywords: MEW; biomimicry; implants; melt electrowriting; tympanic membranes.
© 2021 The Authors. Advanced Healthcare Materials published by Wiley-VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures





Similar articles
-
Multimodal additive manufacturing of biomimetic tympanic membrane replacements with near tissue-like acousto-mechanical and biological properties.Acta Biomater. 2023 Oct 15;170:124-141. doi: 10.1016/j.actbio.2023.09.005. Epub 2023 Sep 9. Acta Biomater. 2023. PMID: 37696412
-
Design, fabrication, and in vitro testing of novel three-dimensionally printed tympanic membrane grafts.Hear Res. 2016 Oct;340:191-203. doi: 10.1016/j.heares.2016.03.005. Epub 2016 Mar 16. Hear Res. 2016. PMID: 26994661 Free PMC article.
-
Parametric control of fiber morphology and tensile mechanics in scaffolds with high aspect ratio geometry produced via melt electrowriting for musculoskeletal soft tissue engineering.J Mech Behav Biomed Mater. 2019 Nov;99:153-160. doi: 10.1016/j.jmbbm.2019.07.013. Epub 2019 Jul 20. J Mech Behav Biomed Mater. 2019. PMID: 31352215
-
Unveiling the potential of melt electrowriting in regenerative dental medicine.Acta Biomater. 2023 Jan 15;156:88-109. doi: 10.1016/j.actbio.2022.01.010. Epub 2022 Jan 10. Acta Biomater. 2023. PMID: 35026478 Free PMC article. Review.
-
Advances in melt electrowriting for cardiovascular applications.Front Bioeng Biotechnol. 2024 Sep 17;12:1425073. doi: 10.3389/fbioe.2024.1425073. eCollection 2024. Front Bioeng Biotechnol. 2024. PMID: 39355277 Free PMC article. Review.
Cited by
-
3D printing of bio-instructive materials: Toward directing the cell.Bioact Mater. 2022 Apr 23;19:292-327. doi: 10.1016/j.bioactmat.2022.04.008. eCollection 2023 Jan. Bioact Mater. 2022. PMID: 35574057 Free PMC article.
-
3D Plotting of Calcium Phosphate Cement and Melt Electrowriting of Polycaprolactone Microfibers in One Scaffold: A Hybrid Additive Manufacturing Process.J Funct Biomater. 2022 Jun 8;13(2):75. doi: 10.3390/jfb13020075. J Funct Biomater. 2022. PMID: 35735931 Free PMC article.
-
In vivo microstructural investigation of the human tympanic membrane by endoscopic polarization-sensitive optical coherence tomography.J Biomed Opt. 2023 Dec;28(12):121203. doi: 10.1117/1.JBO.28.12.121203. Epub 2023 Mar 29. J Biomed Opt. 2023. PMID: 37007626 Free PMC article.
-
Melt Electrowriting of Graded Porous Scaffolds to Mimic the Matrix Structure of the Human Trabecular Meshwork.ACS Biomater Sci Eng. 2022 Sep 12;8(9):3899-3911. doi: 10.1021/acsbiomaterials.2c00623. Epub 2022 Aug 19. ACS Biomater Sci Eng. 2022. PMID: 35984428 Free PMC article.
-
Polyacrylamide/Gel-Based Self-Healing Artificial Tympanic Membrane for Drug Delivery of Otitis Treatment.Biomater Res. 2024 Jul 1;28:0049. doi: 10.34133/bmr.0049. eCollection 2024. Biomater Res. 2024. PMID: 38952716 Free PMC article.
References
-
- Acuin J., W. H. O. Dept. of Child and Adolescent Health and the Team for Prevention of Blindness and Deafness, 2004.
-
- Verhoeff M., van der Veen E. L., Rovers M. M., Sanders E. A. M., Schilder A. G. M., Int. J. Pediatr. Otorhinolaryngol. 2006, 70, 1. - PubMed
-
- Muerbe D., Zahnert T., Bornitz M., Huettenbrink K.‐B., Laryngoscope 2002, 112, 1769. - PubMed
-
- Indorewala S., Laryngoscope 2002, 112, 727. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

