Differential expression of GluN2 NMDA receptor subunits in the dorsal horn of male and female rats
- PMID: 33509021
- PMCID: PMC7849732
- DOI: 10.1080/19336950.2020.1871205
Differential expression of GluN2 NMDA receptor subunits in the dorsal horn of male and female rats
Abstract
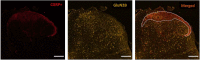
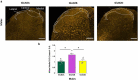
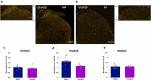
N-methyl-D-aspartate receptors (NMDARs) are excitatory ionotropic glutamate receptors expressed throughout the CNS, including in the spinal dorsal horn. The GluN2 subtypes of NMDAR subunit, which include GluN2A, GluN2B, and GluN2D in the dorsal horn, confer NMDARs with structural and functional variability, enabling heterogeneity in synaptic transmission and plasticity. Despite essential roles for NMDARs in physiological and pathological pain processing, the distribution and function of these specific GluN2 isoforms across dorsal horn laminae remain poorly understood. Surprisingly, there is a complete lack of knowledge of GluN2 expression in female rodents. We, therefore, investigated the relative expression of specific GluN2 variants in the dorsal horn of lumbar (L4/L5) spinal cord from both male and female rats. In order to detect synaptic GluN2 isoforms, we used pepsin antigen-retrieval to unmask these highly cross-linked protein complexes. We found that GluN2B and GluN2D are preferentially localized to the pain-processing superficial regions of the dorsal horn in males, while only GluN2B is predominantly localized to the superficial dorsal horn of female rats. The GluN2A subunit is diffusely localized to neuropil throughout the dorsal horn of both males and females, while GluN2B and GluN2D immunolabelling are found both in the neuropil and on the soma of dorsal horn neurons. Finally, we identified an unexpected enhanced expression of GluN2B in the medial division of the superficial dorsal horn, but in males only. These sex-specific localization patterns of GluN2-NMDAR subunits across dorsal horn laminae have significant implications for the understanding of divergent spinal mechanisms of pain processing.
Keywords: CGRP; GluN2; GluN2A; GluN2B; GluN2D; NMDA receptor; dorsal horn; laminae; sex; spinal cord.
Conflict of interest statement
No potential conflict of interest was reported by the authors.
Figures


Similar articles
-
Heterogeneity of synaptic NMDA receptor responses within individual lamina I pain-processing neurons across sex in rats and humans.J Physiol. 2024 Oct;602(20):5309-5327. doi: 10.1113/JP285521. Epub 2024 Sep 24. J Physiol. 2024. PMID: 39316518
-
Synaptic GluN2A and GluN2B containing NMDA receptors within the superficial dorsal horn activated following primary afferent stimulation.J Neurosci. 2014 Aug 13;34(33):10808-20. doi: 10.1523/JNEUROSCI.0145-14.2014. J Neurosci. 2014. PMID: 25122884 Free PMC article.
-
Conserved contributions of NMDA receptor subtypes to synaptic responses in lamina II spinal neurons across early postnatal development.Mol Brain. 2020 Mar 5;13(1):31. doi: 10.1186/s13041-020-00566-9. Mol Brain. 2020. PMID: 32138769 Free PMC article.
-
GluN2A and GluN2B N-Methyl-D-Aspartate Receptor (NMDARs) Subunits: Their Roles and Therapeutic Antagonists in Neurological Diseases.Pharmaceuticals (Basel). 2023 Oct 30;16(11):1535. doi: 10.3390/ph16111535. Pharmaceuticals (Basel). 2023. PMID: 38004401 Free PMC article. Review.
-
Regulation of NMDA glutamate receptor functions by the GluN2 subunits.J Neurochem. 2020 Jul;154(2):121-143. doi: 10.1111/jnc.14970. Epub 2020 Feb 16. J Neurochem. 2020. PMID: 31978252 Free PMC article. Review.
Cited by
-
Novel GluN2B-Selective NMDA Receptor Negative Allosteric Modulator Possesses Intrinsic Analgesic Properties and Enhances Analgesia of Morphine in a Rodent Tail Flick Pain Model.ACS Chem Neurosci. 2023 Mar 1;14(5):917-935. doi: 10.1021/acschemneuro.2c00779. Epub 2023 Feb 13. ACS Chem Neurosci. 2023. PMID: 36779874 Free PMC article.
-
Understanding the role of the NMDA receptor subunit, GluN2D, in mediating NMDA receptor antagonist-induced behavioral disruptions in male and female mice.J Neurosci Res. 2024 Jan;102(1):e25257. doi: 10.1002/jnr.25257. Epub 2023 Oct 10. J Neurosci Res. 2024. PMID: 37814998 Free PMC article.
-
A review of dorsal root ganglia and primary sensory neuron plasticity mediating inflammatory and chronic neuropathic pain.Neurobiol Pain. 2024 Jan 20;15:100151. doi: 10.1016/j.ynpai.2024.100151. eCollection 2024 Jan-Jun. Neurobiol Pain. 2024. PMID: 38314104 Free PMC article. Review.
-
Advances and Barriers in Understanding Presynaptic N-Methyl-D-Aspartate Receptors in Spinal Pain Processing.Front Mol Neurosci. 2022 Mar 31;15:864502. doi: 10.3389/fnmol.2022.864502. eCollection 2022. Front Mol Neurosci. 2022. PMID: 35431805 Free PMC article. Review.
-
Characterization of Fragile X Mental Retardation Protein expression in human nociceptors and their axonal projections to the spinal dorsal horn.J Comp Neurol. 2023 May;531(7):814-835. doi: 10.1002/cne.25463. Epub 2023 Feb 20. J Comp Neurol. 2023. PMID: 36808110 Free PMC article.
References
-
- Rexed B The cytoarchitectonic organization of the spinal cord in the cat. J Comp Neurol [Internet]. 1952. [cited 2020 December22]; 96:415–95. Available from: https://onlinelibrary.wiley.com/doi/full/10.1002/cne.900960303 - DOI - PubMed
-
- Schoffnegger D, Ruscheweyh R, Sandkühler J. Spread of excitation across modality borders in spinal dorsal horn of neuropathic rats. Pain. 2008;135:300–310. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
