Endothelial cells in the pathogenesis of pulmonary arterial hypertension
- PMID: 33509961
- PMCID: PMC8316496
- DOI: 10.1183/13993003.03957-2020
Endothelial cells in the pathogenesis of pulmonary arterial hypertension
Abstract
Pulmonary arterial hypertension (PAH) is a devastating disease that involves pulmonary vasoconstriction, small vessel obliteration, large vessel thickening and obstruction, and development of plexiform lesions. PAH vasculopathy leads to progressive increases in pulmonary vascular resistance, right heart failure and, ultimately, premature death. Besides other cell types that are known to be involved in PAH pathogenesis (e.g. smooth muscle cells, fibroblasts and leukocytes), recent studies have demonstrated that endothelial cells (ECs) have a crucial role in the initiation and progression of PAH. The EC-specific role in PAH is multi-faceted and affects numerous pathophysiological processes, including vasoconstriction, inflammation, coagulation, metabolism and oxidative/nitrative stress, as well as cell viability, growth and differentiation. In this review, we describe how EC dysfunction and cell signalling regulate the pathogenesis of PAH. We also highlight areas of research that warrant attention in future studies, and discuss potential molecular signalling pathways in ECs that could be targeted therapeutically in the prevention and treatment of PAH.
Copyright ©The authors 2021. For reproduction rights and permissions contact permissions@ersnet.org.
Conflict of interest statement
Conflict of interest: N.D. Cober reports grants from Canadian Institute of Health Research and Canadian Vascular Network, during the conduct of the study. Conflict of interest: Z. Dai reports grants from National Institutes of Health, American Heart Association and American Thoracic Society, during the conduct of the study. Conflict of interest: D.J. Stewart reports other (founding member, equity stake) from Northern Therapeutics, outside the submitted work. Conflict of interest: Y-Y. Zhao reports grants from National Institutes of Health/National Heart, Lung, and Blood Institute (R01HL123957, R01HL133951, R01HL140409 and R01HL148810), during the conduct of the study. Conflict of interest: C.E. Evans reports grants (Career Development Award, 19CDA34500000) from American Heart Association, during the conduct of the study.
Figures

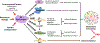
References
-
- Simonneau G, Gatzoulis MA, Adatia I, Celermajer D, Denton C, Ghofrani A, Gomez Sanchez MA, Krishna Kumar R, Landzberg M, Machado RF, Olschewski H, Robbins IM, Souza R. Updated clinical classification of pulmonary hypertension. J Am Coll Cardiol 2013: 62(25 Suppl): D34–41. - PubMed
-
- Rabinovitch M. Pathobiology of pulmonary hypertension. Annu Rev Pathol 2007: 2: 369–399. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
