A randomised, double blind, placebo-controlled trial of megestrol acetate or dexamethasone in treating symptomatic anorexia in people with advanced cancer
- PMID: 33510313
- PMCID: PMC7844230
- DOI: 10.1038/s41598-021-82120-8
A randomised, double blind, placebo-controlled trial of megestrol acetate or dexamethasone in treating symptomatic anorexia in people with advanced cancer
Abstract
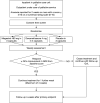
This multi-site, double blind, parallel arm, fixed dose, randomised placebo controlled phase III study compared megestrol acetate 480 mg/day with dexamethasone 4 mg/day for their net effects on appetite in people with cancer anorexia. Patients with advanced cancer and anorexia for ≥ 2 weeks with a score ≤ 4 (0-10 numeric rating scale (NRS) 0 = no appetite, 10 = best possible appetite) were recruited. Participants received megestrol 480 mg or dexamethasone 4 mg or placebo daily for up to 4 weeks. Primary outcomes were at day 7. Responders were defined as having a ≥ 25% improvement in NRS over baseline. There were 190 people randomised (megestrol acetate n = 61; dexamethasone n = 67, placebo n = 62). At week 1 (primary endpoint), 79·3% in the megestrol group, 65·5% in the dexamethasone group and 58·5% in the placebo group (p = 0.067) were responders. No differences in performance status or quality of life were reported. Treatment emergent adverse events were frequent (90·4% of participants), and included altered mood and insomnia. Hyperglycemia and deep vein thromboses were more frequent when on dexamethasone than the other two arms. There was no difference in groups between the three arms, with no benefit seen over placebo with anorexia improving in all arms.Trail registration: The trial was registered on 19/08/2008 with the Australian New Zealand Clinical Trials Registry (ACTRN12608000405314).
Conflict of interest statement
The authors declare no competing interests.
Figures
Similar articles
-
Randomized comparison of megestrol acetate versus dexamethasone versus fluoxymesterone for the treatment of cancer anorexia/cachexia.J Clin Oncol. 1999 Oct;17(10):3299-306. doi: 10.1200/JCO.1999.17.10.3299. J Clin Oncol. 1999. PMID: 10506633 Clinical Trial.
-
Megestrol acetate for anorexia in patients with far-advanced cancer: a double-blind controlled clinical trial.Eur J Cancer. 1998 Oct;34(11):1705-9. doi: 10.1016/s0959-8049(98)00219-6. Eur J Cancer. 1998. PMID: 9893656 Clinical Trial.
-
Anticachectic efficacy of megestrol acetate at different doses and versus placebo in patients with neoplastic cachexia.Am J Clin Oncol. 1998 Aug;21(4):347-51. doi: 10.1097/00000421-199808000-00006. Am J Clin Oncol. 1998. PMID: 9708631 Clinical Trial.
-
Megestrol acetate for cachexia-anorexia syndrome. A systematic review.J Cachexia Sarcopenia Muscle. 2018 Jun;9(3):444-452. doi: 10.1002/jcsm.12292. Epub 2018 Mar 14. J Cachexia Sarcopenia Muscle. 2018. PMID: 29542279 Free PMC article.
-
Treating malnutrition with megestrol acetate: literature review and review of our experience.J Nutr Health Aging. 2002 May;6(3):191-200. J Nutr Health Aging. 2002. PMID: 12152625 Review.
Cited by
-
Role of diet, physical activity and new drugs in the primary management of cancer cachexia in gastrointestinal tumors - a comprehensive review.Front Oncol. 2025 May 30;15:1600425. doi: 10.3389/fonc.2025.1600425. eCollection 2025. Front Oncol. 2025. PMID: 40519290 Free PMC article. Review.
-
Body weight and composition endpoints in cancer cachexia clinical trials: Systematic Review 4 of the cachexia endpoints series.J Cachexia Sarcopenia Muscle. 2024 Jun;15(3):816-852. doi: 10.1002/jcsm.13478. Epub 2024 May 13. J Cachexia Sarcopenia Muscle. 2024. PMID: 38738581 Free PMC article.
-
Appetite Loss in Patients with Advanced Cancer Treated at an Acute Palliative Care Unit.Curr Oncol. 2024 Oct 10;31(10):6061-6072. doi: 10.3390/curroncol31100452. Curr Oncol. 2024. PMID: 39451756 Free PMC article.
-
Effects of Reflexology on Anorexia and Sleep Quality in Patients Undergoing Chemotherapy: A Randomized Clinical Trial.Iran J Nurs Midwifery Res. 2024 Jul 2;29(3):337-342. doi: 10.4103/ijnmr.ijnmr_62_23. eCollection 2024 May-Jun. Iran J Nurs Midwifery Res. 2024. PMID: 39100404 Free PMC article.
-
A taste alteration-related scale assesses megestrol to improve chemotherapy-induced anorexia.Support Care Cancer. 2024 Apr 22;32(5):300. doi: 10.1007/s00520-024-08499-y. Support Care Cancer. 2024. PMID: 38644409
References
-
- Dunlop R. Clinical epidemiology of cancer cachexia. In: Bruera E, Higginson I, editors. Cachexia-Anorexia in Cancer Patients. Oxford: Oxford University Press; 1996. pp. 76–82.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

