Inhibitors of Src Family Kinases, Inducible Nitric Oxide Synthase, and NADPH Oxidase as Potential CNS Drug Targets for Neurological Diseases
- PMID: 33515429
- PMCID: PMC7893831
- DOI: 10.1007/s40263-020-00787-5
Inhibitors of Src Family Kinases, Inducible Nitric Oxide Synthase, and NADPH Oxidase as Potential CNS Drug Targets for Neurological Diseases
Abstract
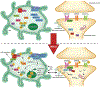
Neurological diseases share common neuroinflammatory and oxidative stress pathways. Both phenotypic and molecular changes in microglia, astrocytes, and neurons contribute to the progression of disease and present potential targets for disease modification. Src family kinases (SFKs) are present in both neurons and glial cells and are upregulated following neurological insults in both human and animal models. In neurons, SFKs interact with post-synaptic protein domains to mediate hyperexcitability and neurotoxicity. SFKs are upstream of signaling cascades that lead to the modulation of neurotransmitter receptors and the transcription of pro-inflammatory cytokines as well as producers of free radicals through the activation of glia. Inducible nitric oxide synthase (iNOS/NOS-II) and nicotinamide adenine dinucleotide phosphate (NADPH) oxidase 2 (NOX2), the major mediators of reactive nitrogen/oxygen species (RNS/ROS) production in the brain, are also upregulated along with the pro-inflammatory cytokines following neurological insult and contribute to disease progression. Persistent neuronal hyperexcitability, RNS/ROS, and cytokines can exacerbate neurodegeneration, a common pathognomonic feature of the most prevalent neurological disorders such as Alzheimer's disease, Parkinson's disease, and epilepsy. Using a wide variety of preclinical disease models, inhibitors of the SFK-iNOS-NOX2 signaling axis have been tested to cure or modify disease progression. In this review, we discuss the SFK-iNOS-NOX2 signaling pathway and their inhibitors as potential CNS targets for major neurological diseases.
Trial registration: ClinicalTrials.gov NCT00992667 NCT04327089.
Plain language summary
Nerve cell death, oxidative stress, and inflammation of the brain are the most common pathological processes of many neurological diseases. These processes are mediated through changes in glial cells, the supporting cells in the brain, via several molecular pathways. Some of these pathways are potential drug targets for the mitigation of brain pathology. In this review, we focus on pathways involving Src family kinases, inducible nitric oxide synthase and nicotinamide adenine dinucleotide phosphate oxidase, and their inhibitors, which are promising agents for modifying neurological diseases.
Conflict of interest statement
Conflicts of Interest
The authors report no conflicts of interest
Figures


References
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

