Development of a Novel Chimeric Endolysin, Lys109 With Enhanced Lytic Activity Against Staphylococcus aureus
- PMID: 33519773
- PMCID: PMC7843465
- DOI: 10.3389/fmicb.2020.615887
Development of a Novel Chimeric Endolysin, Lys109 With Enhanced Lytic Activity Against Staphylococcus aureus
Abstract
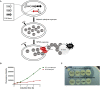
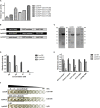
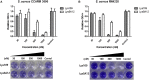
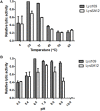
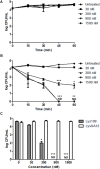
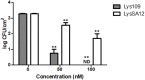
As the incidence of antibiotic-resistant bacteria has become increased, phage endolysins are believed as one of the promising alternatives to antibiotics. However, the discovery of potent endolysin is still challenging because it is labor intensive and difficult to obtain a soluble form with high lytic activity. In this respect, the modular structures of Gram-positive endolysins can provide an opportunity to develop novel endolysins by domain rearrangement. In this study, a random domain swapping library of four different endolysins from phages infecting Staphylococcus aureus was constructed and screened to obtain engineered endolysins. The novel chimeric endolysin, Lys109 was selected and characterized for its staphylolytic activity. Lys109 exhibited greater bacterial cell lytic activity than its parental endolysins against staphylococcal planktonic cells and biofilms, showing highly improved activity in eliminating S. aureus from milk and on the surface of stainless steel. These results demonstrate that a novel chimeric endolysin with higher activity and solubility can be developed by random domain swapping and that this chimeric endolysin has a great potential as an antimicrobial agent.
Keywords: Staphylococcus aureus; antimicrobial agent; domain swapping; endolysin; screening.
Copyright © 2021 Son, Kong, Lee and Ryu.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Similar articles
-
Development of Advanced Chimeric Endolysin to Control Multidrug-Resistant Staphylococcus aureus through Domain Shuffling.ACS Infect Dis. 2021 Aug 13;7(8):2081-2092. doi: 10.1021/acsinfecdis.0c00812. Epub 2021 May 28. ACS Infect Dis. 2021. PMID: 34047546
-
Design, Screening, and Characterization of Engineered Phage Endolysins with Extracellular Antibacterial Activity against Gram-Negative Bacteria.Appl Environ Microbiol. 2023 Jul 26;89(7):e0058123. doi: 10.1128/aem.00581-23. Epub 2023 Jun 20. Appl Environ Microbiol. 2023. PMID: 37338346 Free PMC article.
-
Simultaneous Control of Staphylococcus aureus and Bacillus cereus Using a Hybrid Endolysin LysB4EAD-LysSA11.Antibiotics (Basel). 2020 Dec 14;9(12):906. doi: 10.3390/antibiotics9120906. Antibiotics (Basel). 2020. PMID: 33327470 Free PMC article.
-
The Synergistic and Chimeric Mechanism of Bacteriophage Endolysins: Opportunities for Application in Biotherapeutics, Food, and Health Sectors.Probiotics Antimicrob Proteins. 2025 Apr;17(2):807-831. doi: 10.1007/s12602-024-10394-1. Epub 2024 Nov 7. Probiotics Antimicrob Proteins. 2025. PMID: 39508962 Review.
-
Recombinant Endolysins as Potential Therapeutics against Antibiotic-Resistant Staphylococcus aureus: Current Status of Research and Novel Delivery Strategies.Clin Microbiol Rev. 2017 Nov 29;31(1):e00071-17. doi: 10.1128/CMR.00071-17. Print 2018 Jan. Clin Microbiol Rev. 2017. PMID: 29187396 Free PMC article. Review.
Cited by
-
Treating Bacterial Infections with Bacteriophage-Based Enzybiotics: In Vitro, In Vivo and Clinical Application.Antibiotics (Basel). 2021 Dec 6;10(12):1497. doi: 10.3390/antibiotics10121497. Antibiotics (Basel). 2021. PMID: 34943709 Free PMC article. Review.
-
Phage-derived proteins: Advancing food safety through biocontrol and detection of foodborne pathogens.Compr Rev Food Sci Food Saf. 2025 Mar;24(2):e70124. doi: 10.1111/1541-4337.70124. Compr Rev Food Sci Food Saf. 2025. PMID: 39898971 Free PMC article. Review.
-
Synergistic action of phages and lytic proteins with antibiotics: a combination strategy to target bacteria and biofilms.BMC Microbiol. 2023 May 23;23(1):149. doi: 10.1186/s12866-023-02881-2. BMC Microbiol. 2023. PMID: 37221517 Free PMC article.
-
Lysin and Lytic Phages Reduce Vibrio Counts in Live Feed and Fish Larvae.Microorganisms. 2024 Apr 30;12(5):904. doi: 10.3390/microorganisms12050904. Microorganisms. 2024. PMID: 38792735 Free PMC article.
-
Development of engineered endolysins with in vitro intracellular activity against streptococcal bovine mastitis-causing pathogens.Microb Biotechnol. 2023 Dec;16(12):2367-2386. doi: 10.1111/1751-7915.14339. Epub 2023 Oct 18. Microb Biotechnol. 2023. PMID: 37853918 Free PMC article.
References
-
- Becker S. C., Foster-Frey J., Stodola A. J., Anacker D., Donovan D. M. (2009b). Differentially conserved staphylococcal SH3b_5 cell wall binding domains confer increased staphylolytic and streptolytic activity to a streptococcal prophage endolysin domain. Gene 443 32–41. 10.1016/j.gene.2009.04.023 - DOI - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

