Catalytic hydrogen atom transfer to alkenes: a roadmap for metal hydrides and radicals
- PMID: 33520153
- PMCID: PMC7810138
- DOI: 10.1039/d0sc04112b
Catalytic hydrogen atom transfer to alkenes: a roadmap for metal hydrides and radicals
Abstract
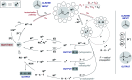
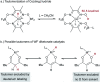
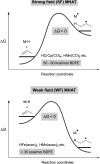
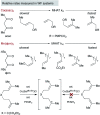
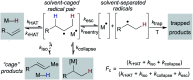
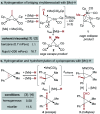
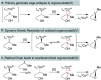
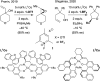
Hydrogen atom transfer from a metal hydride (MHAT) has emerged as a powerful, if puzzling, technique in chemical synthesis. In catalytic MHAT reactions, earth-abundant metal complexes generate stabilized and unstabilized carbon-centered radicals from alkenes of various substitution patterns with robust chemoselectivity. This perspective combines organic and inorganic perspectives to outline challenges and opportunities, and to propose working models to assist further developments. We attempt to demystify the putative intermediates, the basic elementary steps, and the energetic implications, especially for cage pair formation, collapse and separation. Distinctions between catalysts with strong-field (SF) and weak-field (WF) ligand environments may explain some differences in reactivity and selectivity, and provide an organizing principle for kinetics that transcends the typical thermodynamic analysis. This blueprint should aid practitioners who hope to enter and expand this exciting area of chemistry.
This journal is © The Royal Society of Chemistry 2020.
Figures

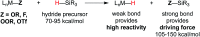



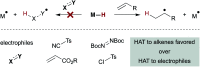









Similar articles
-
The High Chemofidelity of Metal-Catalyzed Hydrogen Atom Transfer.Acc Chem Res. 2018 Nov 20;51(11):2628-2640. doi: 10.1021/acs.accounts.8b00337. Epub 2018 Nov 8. Acc Chem Res. 2018. PMID: 30406655 Free PMC article.
-
Intercepting Hydrogen Evolution with Hydrogen-Atom Transfer: Electron-Initiated Hydrofunctionalization of Alkenes.J Am Chem Soc. 2022 Oct 5;144(39):17783-17791. doi: 10.1021/jacs.2c08278. Epub 2022 Sep 22. J Am Chem Soc. 2022. PMID: 36137298
-
Manganese-Oxygen Intermediates in O-O Bond Activation and Hydrogen-Atom Transfer Reactions.Acc Chem Res. 2017 Nov 21;50(11):2706-2717. doi: 10.1021/acs.accounts.7b00343. Epub 2017 Oct 24. Acc Chem Res. 2017. PMID: 29064667 Review.
-
Unraveling Hydrogen Atom Transfer Mechanisms with Voltammetry: Oxidative Formation and Reactivity of Cobalt Hydride.J Am Chem Soc. 2023 Aug 16;145(32):17665-17677. doi: 10.1021/jacs.3c03815. Epub 2023 Aug 2. J Am Chem Soc. 2023. PMID: 37530748
-
Photo- and Electrochemical Cobalt Catalysed Hydrogen Atom Transfer for the Hydrofunctionalisation of Alkenes.Angew Chem Int Ed Engl. 2023 Oct 9;62(41):e202304882. doi: 10.1002/anie.202304882. Epub 2023 Jul 11. Angew Chem Int Ed Engl. 2023. PMID: 37184388 Review.
Cited by
-
Interplay between β-Diimino and β-Diketiminato Ligands in Nickel Complexes Active in the Proton Reduction Reaction.Inorg Chem. 2022 Oct 24;61(42):16639-16649. doi: 10.1021/acs.inorgchem.2c02150. Epub 2022 Oct 5. Inorg Chem. 2022. PMID: 36196853 Free PMC article.
-
Photocatalytic phosphine-mediated water activation for radical hydrogenation.Nature. 2023 Jul;619(7970):506-513. doi: 10.1038/s41586-023-06141-1. Epub 2023 Jun 28. Nature. 2023. PMID: 37380779 Free PMC article.
-
Mechanistic Studies on the Hexadecafluorophthalocyanine-Iron-Catalyzed Wacker-Type Oxidation of Olefins to Ketones*.Chemistry. 2021 Dec 1;27(67):16776-16787. doi: 10.1002/chem.202102848. Epub 2021 Oct 27. Chemistry. 2021. PMID: 34546596 Free PMC article.
-
Metal-Hydride C-C Cross-Coupling of Alkenes Through a Double Outer-Sphere Mechanism.J Org Chem. 2024 Nov 15;89(22):16106-16113. doi: 10.1021/acs.joc.4c00260. Epub 2024 Jun 26. J Org Chem. 2024. PMID: 38926670 Review.
-
Intermolecular Hydroalkoxylation and Hydrocarboxylation of 2-Azadienes with High Efficiency.J Org Chem. 2023 Mar 3;88(5):3277-3281. doi: 10.1021/acs.joc.2c02534. Epub 2023 Feb 21. J Org Chem. 2023. PMID: 36802598 Free PMC article.
References
-
- Hartwig J. F., Organotransition Metal Chemistry, University Science Books, Sausalito, CA, 2010.
-
- Holland P. L. Acc. Chem. Res. 2015;48:1696–1702. - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

