Tonic GABAergic inhibition, via GABAA receptors containing αβƐ subunits, regulates excitability of ventral tegmental area dopamine neurons
- PMID: 33522050
- PMCID: PMC8651010
- DOI: 10.1111/ejn.15133
Tonic GABAergic inhibition, via GABAA receptors containing αβƐ subunits, regulates excitability of ventral tegmental area dopamine neurons
Abstract
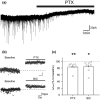
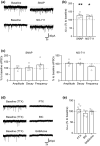
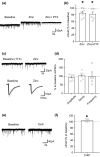
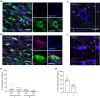
The activity of midbrain dopamine neurons is strongly regulated by fast synaptic inhibitory γ-Aminobutyric acid (GABA)ergic inputs. There is growing evidence in other brain regions that low concentrations of ambient GABA can persistently activate certain subtypes of GABAA receptor to generate a tonic current. However, evidence for a tonic GABAergic current in midbrain dopamine neurons is limited. To address this, we conducted whole-cell recordings from ventral tegmental area (VTA) dopamine neurons in brain slices from mice. We found that application of GABAA receptor antagonists decreased the holding current, indicating the presence of a tonic GABAergic input. Global increases in GABA release, induced by either a nitric oxide donor or inhibition of GABA uptake, further increased this tonic current. Importantly, prolonged inhibition of the firing activity of local GABAergic neurons abolished the tonic current. A combination of pharmacology and immunohistochemistry experiments suggested that, unlike common examples of tonic inhibition, this current may be mediated by a relatively unusual combination of α4βƐ subunits. Lastly, we found that the tonic current reduced excitability in dopamine neurons suggesting a subtractive effect on firing activity.
Keywords: addiction; extra-synaptic; midbrain; synaptic transmission.
© 2021 The Authors. European Journal of Neuroscience published by Federation of European Neuroscience Societies and John Wiley & Sons Ltd.
Conflict of interest statement
The authors declare no competing financial interests.
Figures
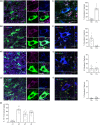
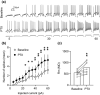
Similar articles
-
Ethanol dually modulates GABAergic synaptic transmission onto dopaminergic neurons in ventral tegmental area: role of mu-opioid receptors.Neuroscience. 2008 Apr 22;153(1):240-8. doi: 10.1016/j.neuroscience.2008.01.040. Epub 2008 Feb 6. Neuroscience. 2008. PMID: 18343590 Free PMC article.
-
Activation of LH GABAergic inputs counteracts fasting-induced changes in tVTA/RMTG neurons.J Physiol. 2022 May;600(9):2203-2224. doi: 10.1113/JP282653. Epub 2022 Apr 13. J Physiol. 2022. PMID: 35338656
-
Ethanol enhances GABAergic transmission onto dopamine neurons in the ventral tegmental area of the rat.Alcohol Clin Exp Res. 2008 Jun;32(6):1040-8. doi: 10.1111/j.1530-0277.2008.00665.x. Epub 2008 Apr 15. Alcohol Clin Exp Res. 2008. PMID: 18422836 Free PMC article.
-
GABAergic neurons in the ventral tegmental area receive dual GABA/enkephalin-mediated inhibitory inputs from the bed nucleus of the stria terminalis.Eur J Neurosci. 2014 Jun;39(11):1796-809. doi: 10.1111/ejn.12503. Epub 2014 Mar 2. Eur J Neurosci. 2014. PMID: 24580812
-
Synaptic function and plasticity in identified inhibitory inputs onto VTA dopamine neurons.Eur J Neurosci. 2018 May;47(10):1208-1218. doi: 10.1111/ejn.13879. Epub 2018 Mar 13. Eur J Neurosci. 2018. PMID: 29480954 Free PMC article.
Cited by
-
Axonal Modulation of Striatal Dopamine Release by Local γ-Aminobutyric Acid (GABA) Signalling.Cells. 2021 Mar 23;10(3):709. doi: 10.3390/cells10030709. Cells. 2021. PMID: 33806845 Free PMC article. Review.
-
GABA co-released from striatal dopamine axons dampens phasic dopamine release through autoregulatory GABAA receptors.Cell Rep. 2024 Mar 26;43(3):113834. doi: 10.1016/j.celrep.2024.113834. Epub 2024 Mar 2. Cell Rep. 2024. PMID: 38431842 Free PMC article.
-
Chemogenetic Modulation of Preoptic Gabre Neurons Decreases Body Temperature and Heart Rate.Int J Mol Sci. 2024 Dec 5;25(23):13061. doi: 10.3390/ijms252313061. Int J Mol Sci. 2024. PMID: 39684772 Free PMC article.
-
GABA tone regulation and its cognitive functions in the brain.Nat Rev Neurosci. 2023 Sep;24(9):523-539. doi: 10.1038/s41583-023-00724-7. Epub 2023 Jul 26. Nat Rev Neurosci. 2023. PMID: 37495761 Review.
-
Dopamine transporter blockade during adolescence increases adult dopamine function, impulsivity, and aggression.Mol Psychiatry. 2023 Aug;28(8):3512-3523. doi: 10.1038/s41380-023-02194-w. Epub 2023 Aug 2. Mol Psychiatry. 2023. PMID: 37532798 Free PMC article.
References
-
- Belujon, P. , Baufreton, J. , Grandoso, L. , Boué‐Grabot, E. , Batten, T. F. C. , Ugedo, L. , Garret, M. , & Taupignon, A. I. (2009). Inhibitory transmission in locus coeruleus neurons expressing GABAA receptor epsilon subunit has a number of unique properties. Journal of Neurophysiology, 102, 2312–2325. - PubMed
-
- Bonnert, T. P. , McKernan, R. M. , Farrar, S. , le Bourdellès, B. , Heavens, R. P. , Smith, D. W. , Hewson, L. , Rigby, M. R. , Sirinathsinghji, D. J. , Brown, N. , Wafford, K. A. , & Whiting, P. J. (1999). theta, a novel gamma‐aminobutyric acid type A receptor subunit. Proceedings of the National Academy of Sciences of the United States of America, 96, 9891–9896. 10.1073/pnas.96.17.9891 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

