Encapsulated VEGF121-PLA microparticles promote angiogenesis in human endometrium stromal cells
- PMID: 33523322
- PMCID: PMC7851192
- DOI: 10.1186/s43141-021-00118-1
Encapsulated VEGF121-PLA microparticles promote angiogenesis in human endometrium stromal cells
Abstract
Background: In this study, Vascular Endothelial Growth Factor 121 expressed abundantly in endometrial stromal cells is encapsulated with poly-l-lactide and characterized the properties for endometrial angiogenesis. We studied the migration, proliferation and the protein levels of human immortalized endometrium stromal cells after treating the cells with recombinant Vascular Endothelial Growth Factor (200 and 500 nanogram), and poly-l-lactide loaded Vascular Endothelial Growth Factor 121 (day 1, 20 and 30). The present study explains endometrium angiogenesis because endometrium plays an important role in pregnancy.
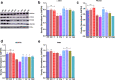
Results: Migration and proliferation studies in endometrium cells proved the efficiency of Vascular Endothelial Growth Factor and poly-l-lactide loaded Vascular Endothelial Growth Factor 121. This proliferated and increased the migration of the cells in vitro and also activated the Protein kinase B, Phosphatidylinositol-4, 5-Bisphosphate 3-Kinase Catalytic Subunit Beta, α-Smooth muscle actin and vascular endothelial growth factor receptor 2 pathways. Western blot analysis showed the increased expression levels of kinases, smooth muscle actin and vascular endothelial growth factor receptor 2 after the treatment with Vascular Endothelial Growth Factor and poly-l-lactide loaded Vascular Endothelial Growth Factor 121 particles in comparison to the control group. The elevated levels of α-Smooth muscle actin in endometrium cells with Vascular Endothelial Growth Factor prove the regulation of angiogenesis in vitro.
Conclusion: Endometrium thickness is one of the important factors during implantation of embryo and pregnancy. Slow release of VEGF from PLA encapsulated microparticle further controls the endothelial cell proliferation and migration and helps in the promotion of angiogenesis. The combined effect studied in vitro could be used as a pro-angiogenic drug on further in vivo confirmation.
Keywords: Angiogenesis; Endometrium; Human endometrium stromal cells; Migration; Proliferation; VEGF.
Conflict of interest statement
The authors state that they do not have any conflict of interest.
Figures



References
-
- Sharkey AM, Day K, McPherson A, Malik S, Licence D, Smith SK, et al. Vascular endothelial growth factor expression in human endometrium is regulated by hypoxia. J Clin Endocrinol Metab. 2000;85:402–409. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
