Open-Label, Single-Arm, Phase II Study of Pembrolizumab Monotherapy as First-Line Therapy in Patients With Advanced Non-Clear Cell Renal Cell Carcinoma
- PMID: 33529058
- PMCID: PMC8078262
- DOI: 10.1200/JCO.20.02365
Open-Label, Single-Arm, Phase II Study of Pembrolizumab Monotherapy as First-Line Therapy in Patients With Advanced Non-Clear Cell Renal Cell Carcinoma
Abstract
Purpose: Programmed death 1 (PD-1) pathway inhibitors have not been prospectively evaluated in patients with non-clear cell renal cell carcinoma (nccRCC). The phase II KEYNOTE-427 study (cohort B) was conducted to assess the efficacy and safety of single-agent pembrolizumab, a PD-1 inhibitor, in advanced nccRCC.
Methods: Patients with histologically confirmed, measurable (Response Evaluation Criteria in Solid Tumors [RECIST] version 1.1) nccRCC and no prior systemic therapy received pembrolizumab 200 mg intravenously once every 3 weeks for ≤ 24 months. The primary end point was objective response rate (ORR) per RECIST v1.1.
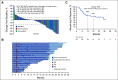
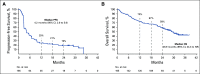
Results: Among enrolled patients (N = 165), 71.5% had confirmed papillary, 12.7% had chromophobe, and 15.8% had unclassified RCC histology. Most patients (67.9%) had intermediate or poor International Metastatic RCC Database Consortium risk status and tumors with programmed death ligand 1 (PD-L1) combined positive score (CPS) ≥ 1 (61.8%). The median time from enrollment to database cutoff was 31.5 months (range, 22.7-38.8). In all patients, the ORR was 26.7%. The median duration of response was 29.0 months; 59.7% of responses lasted ≥ 12 months. The ORR by CPS ≥ 1 and CPS < 1 status was 35.3% and 12.1%, respectively. The ORR by histology was 28.8% for papillary, 9.5% for chromophobe, and 30.8% for unclassified. Overall, the median progression-free survival was 4.2 months (95% CI, 2.9 to 5.6); the 24-month rate was 18.6%. The median overall survival was 28.9 months (95% CI, 24.3 months to not reached); the 24-month rate was 58.4%. Overall, 69.7% of patients reported treatment-related adverse events, most commonly pruritus (20.0%) and hypothyroidism (14.5%). Two deaths were treatment related (pneumonitis and cardiac arrest).
Conclusion: First-line pembrolizumab monotherapy showed promising antitumor activity in nccRCC. The safety profile was similar to that observed in other tumor types.
Trial registration: ClinicalTrials.gov NCT02853344.
Figures

References
-
- National Comprehensive Cancer Network : NCCN Clinical Practice Guidelines in Oncology-Kidney Cancer. V.1.2021, National Comprehensive Cancer Network, 2020. https://www.nccn.org/professionals/physician_gls/pdf/kidney.pdf - PubMed
-
- Zhang T Gong J Maia MC, et al. : Systemic therapy for non-clear cell renal cell carcinoma. Am Soc Clin Oncol Educ Book 37:337-342, 2017 - PubMed
-
- Motzer RJ Bacik J Mariani T, et al. : Treatment outcome and survival associated with metastatic renal cell carcinoma of non-clear-cell histology. J Clin Oncol 20:2376-2381, 2002 - PubMed
-
- Motzer RJ Bacik J Murphy BA, et al. : Interferon-alfa as a comparative treatment for clinical trials of new therapies against advanced renal cell carcinoma. J Clin Oncol 20:289-296, 2002 - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

