Tsetse salivary glycoproteins are modified with paucimannosidic N-glycans, are recognised by C-type lectins and bind to trypanosomes
- PMID: 33529215
- PMCID: PMC7880456
- DOI: 10.1371/journal.pntd.0009071
Tsetse salivary glycoproteins are modified with paucimannosidic N-glycans, are recognised by C-type lectins and bind to trypanosomes
Abstract
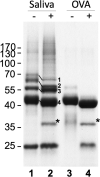
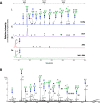
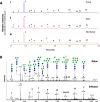
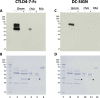
African sleeping sickness is caused by Trypanosoma brucei, a parasite transmitted by the bite of a tsetse fly. Trypanosome infection induces a severe transcriptional downregulation of tsetse genes encoding for salivary proteins, which reduces its anti-hemostatic and anti-clotting properties. To better understand trypanosome transmission and the possible role of glycans in insect bloodfeeding, we characterized the N-glycome of tsetse saliva glycoproteins. Tsetse salivary N-glycans were enzymatically released, tagged with either 2-aminobenzamide (2-AB) or procainamide, and analyzed by HILIC-UHPLC-FLR coupled online with positive-ion ESI-LC-MS/MS. We found that the N-glycan profiles of T. brucei-infected and naïve tsetse salivary glycoproteins are almost identical, consisting mainly (>50%) of highly processed Man3GlcNAc2 in addition to several other paucimannose, high mannose, and few hybrid-type N-glycans. In overlay assays, these sugars were differentially recognized by the mannose receptor and DC-SIGN C-type lectins. We also show that salivary glycoproteins bind strongly to the surface of transmissible metacyclic trypanosomes. We suggest that although the repertoire of tsetse salivary N-glycans does not change during a trypanosome infection, the interactions with mannosylated glycoproteins may influence parasite transmission into the vertebrate host.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures


Similar articles
-
Tsetse fly tolerance to T. brucei infection: transcriptome analysis of trypanosome-associated changes in the tsetse fly salivary gland.BMC Genomics. 2016 Nov 25;17(1):971. doi: 10.1186/s12864-016-3283-0. BMC Genomics. 2016. PMID: 27884110 Free PMC article.
-
Tsetse Fly Transmission Studies of African Trypanosomes.Methods Mol Biol. 2020;2116:49-67. doi: 10.1007/978-1-0716-0294-2_4. Methods Mol Biol. 2020. PMID: 32221913
-
Trypanosoma brucei modifies the tsetse salivary composition, altering the fly feeding behavior that favors parasite transmission.PLoS Pathog. 2010 Jun 3;6(6):e1000926. doi: 10.1371/journal.ppat.1000926. PLoS Pathog. 2010. PMID: 20532213 Free PMC article.
-
Flying tryps: survival and maturation of trypanosomes in tsetse flies.Trends Parasitol. 2013 Apr;29(4):188-96. doi: 10.1016/j.pt.2013.02.003. Epub 2013 Mar 16. Trends Parasitol. 2013. PMID: 23507033 Review.
-
Antigenic variation during the developmental cycle of Trypanosoma brucei.J Protozool. 1984 Feb;31(1):41-7. J Protozool. 1984. PMID: 6204043 Review.
Cited by
-
Genetic variation and microbiota in bumble bees cross-infected by different strains of C. bombi.PLoS One. 2022 Nov 28;17(11):e0277041. doi: 10.1371/journal.pone.0277041. eCollection 2022. PLoS One. 2022. PMID: 36441679 Free PMC article.
References
-
- Giunchetti RC, Correa-Oliveira R, Martins-Filho OA, Teixeira-Carvalho A, Roatt BM, de Oliveira Aguiar-Soares RD, et al. A killed Leishmania vaccine with sand fly saliva extract and saponin adjuvant displays immunogenicity in dogs. Vaccine. 2008;26(5):623–38. Epub 2008/01/09. 10.1016/j.vaccine.2007.11.057 . - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

