Poor Corticospinal Motor Neuron Health Is Associated with Increased Symptom Severity in the Acute Phase Following Repetitive Mild TBI and Predicts Early ALS Onset in Genetically Predisposed Rodents
- PMID: 33530492
- PMCID: PMC7911729
- DOI: 10.3390/brainsci11020160
Poor Corticospinal Motor Neuron Health Is Associated with Increased Symptom Severity in the Acute Phase Following Repetitive Mild TBI and Predicts Early ALS Onset in Genetically Predisposed Rodents
Abstract
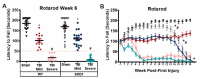
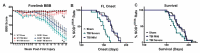
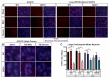
Traumatic brain injury (TBI) is a well-established risk factor for several neurodegenerative disorders including Alzheimer's disease and Parkinson's disease, however, a link between TBI and amyotrophic lateral sclerosis (ALS) has not been clearly elucidated. Using the SOD1G93A rat model known to recapitulate the human ALS condition, we found that exposure to mild, repetitive TBI lead ALS rats to experience earlier disease onset and shortened survival relative to their sham counterparts. Importantly, increased severity of early injury symptoms prior to the onset of ALS disease symptoms was linked to poor health of corticospinal motor neurons and predicted worsened outcome later in life. Whereas ALS rats with only mild behavioral injury deficits exhibited no observable changes in corticospinal motor neuron health and did not present with early onset or shortened survival, those with more severe injury-related deficits exhibited alterations in corticospinal motor neuron health and presented with significantly earlier onset and shortened lifespan. While these studies do not imply that TBI causes ALS, we provide experimental evidence that head injury is a risk factor for earlier disease onset in a genetically predisposed ALS population and is associated with poor health of corticospinal motor neurons.
Keywords: amyotrophic lateral sclerosis; chronic traumatic encephalopathy; concussion; corticospinal motor neurons; traumatic brain injury.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
-
- Rosen D.R., Siddique T., Patterson D., Figlewicz D.A., Sapp P.C., Hentati A., Donaldson D.H., Goto J., O’Regan J.P., Deng H.-X., et al. Mutations in Cu/Zn superoxide dismutase gene are associated with familial amyotrophic lateral sclerosis. Nature. 1993;362:59–62. doi: 10.1038/362059a0. - DOI - PubMed
-
- Ozdinler P.H., Benn S., Yamamoto T.H., Güzel M., Brown R.H., Macklis J.D. Corticospinal Motor Neurons and Related Subcerebral Projection Neurons Undergo Early and Specific Neurodegeneration in hSOD1G93A Transgenic ALS Mice. J. Neurosci. 2011;31:4166–4177. doi: 10.1523/JNEUROSCI.4184-10.2011. - DOI - PMC - PubMed
-
- Thomsen G.M., Gowing G., Latter J., Chen M., Vit J.-P., Staggenborg K., Avalos P., Alkaslasi M., Ferraiuolo L., Likhite S., et al. Delayed Disease Onset and Extended Survival in the SOD1G93A Rat Model of Amyotrophic Lateral Sclerosis after Suppression of Mutant SOD1 in the Motor Cortex. J. Neurosci. 2014;34:15587–15600. doi: 10.1523/JNEUROSCI.2037-14.2014. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

