PON2 subverts metabolic gatekeeper functions in B cells to promote leukemogenesis
- PMID: 33531346
- PMCID: PMC7896313
- DOI: 10.1073/pnas.2016553118
PON2 subverts metabolic gatekeeper functions in B cells to promote leukemogenesis
Abstract
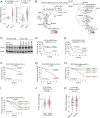
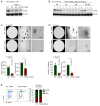
Unlike other cell types, developing B cells undergo multiple rounds of somatic recombination and hypermutation to evolve high-affinity antibodies. Reflecting the high frequency of DNA double-strand breaks, adaptive immune protection by B cells comes with an increased risk of malignant transformation. B lymphoid transcription factors (e.g., IKZF1 and PAX5) serve as metabolic gatekeepers by limiting glucose to levels insufficient to fuel transformation. We here identified aberrant expression of the lactonase PON2 in B cell acute lymphoblastic leukemia (B-ALL) as a mechanism to bypass metabolic gatekeeper functions. Compared to normal pre-B cells, PON2 expression was elevated in patient-derived B-ALL samples and correlated with poor clinical outcomes in pediatric and adult cohorts. Genetic deletion of Pon2 had no measurable impact on normal B cell development. However, in mouse models for BCR-ABL1 and NRASG12D-driven B-ALL, deletion of Pon2 compromised proliferation, colony formation, and leukemia initiation in transplant recipient mice. Compromised leukemogenesis resulted from defective glucose uptake and adenosine triphosphate (ATP) production in PON2-deficient murine and human B-ALL cells. Mechanistically, PON2 enabled glucose uptake by releasing the glucose-transporter GLUT1 from its inhibitor stomatin (STOM) and genetic deletion of STOM largely rescued PON2 deficiency. While not required for glucose transport, the PON2 lactonase moiety hydrolyzes the lactone-prodrug 3OC12 to form a cytotoxic intermediate. Mirroring PON2 expression levels in B-ALL, 3OC12 selectively killed patient-derived B-ALL cells but was well tolerated in transplant recipient mice. Hence, while B-ALL cells critically depend on aberrant PON2 expression to evade metabolic gatekeeper functions, PON2 lactonase activity can be leveraged as synthetic lethality to overcome drug resistance in refractory B-ALL.
Keywords: B cell leukemia; glucose transport; lactonase; paraoxonase 2.
Conflict of interest statement
The authors declare no competing interest.
Figures




References
-
- Buchner M., Swaminathan S., Chen Z., Müschen M., Mechanisms of pre-B-cell receptor checkpoint control and its oncogenic subversion in acute lymphoblastic leukemia. Immunol. Rev. 263, 192–209 (2015). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

