SIRT6 inhibits metastasis by suppressing SNAIL expression in nasopharyngeal carcinoma cells
- PMID: 33532024
- PMCID: PMC7847487
SIRT6 inhibits metastasis by suppressing SNAIL expression in nasopharyngeal carcinoma cells
Abstract
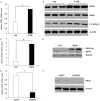
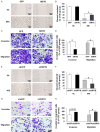
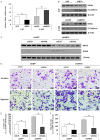
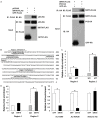
Nasopharyngeal carcinoma (NPC) is a head and neck cancer with severe local invasion and early distant metastasis. SIRT6 serves as a critical modulator of the development and metastasis of multiple types of cancer; however, the roles and underlying mechanisms of SIRT6 in regulating NPC metastasis remain largely unknown. Here, the expression of SIRT6 in high metastatic 5-8F cells and low metastatic 6-10B cells was analyzed. SIRT6 expression was found to be negatively associated with the metastatic capability of NPC cells. Moreover, we identified that SIRT6 inhibited NPC cell metastasis through suppression of SNAIL expression. Mechanistically, we demonstrated that SIRT6 interacted with transcription factor p65 (NF-kB subunit) and deacetylated histone H3 lysine 9 (H3K9) and lysine 56 (H3K56) at the promoter of SNAIL, leading to reduced transcription of SNAIL. In summary, SIRT6 functions as a metastasis suppressor in NPC cells through epigenetic regulation of SNAIL gene expression.
Keywords: Nasopharyngeal carcinoma; SIRT6; SNAIL; metastasis; p65.
IJCEP Copyright © 2021.
Conflict of interest statement
None.
Figures
Similar articles
-
HOPX hypermethylation promotes metastasis via activating SNAIL transcription in nasopharyngeal carcinoma.Nat Commun. 2017 Feb 1;8:14053. doi: 10.1038/ncomms14053. Nat Commun. 2017. PMID: 28146149 Free PMC article.
-
Snail promotes metastasis of nasopharyngeal carcinoma partly by down-regulating TEL2.Cancer Commun (Lond). 2018 Sep 25;38(1):58. doi: 10.1186/s40880-018-0328-6. Cancer Commun (Lond). 2018. PMID: 30253797 Free PMC article.
-
H19/miR-675-5p Targeting SFN Enhances the Invasion and Metastasis of Nasalpharyngeal Cancer Cells.Curr Mol Pharmacol. 2019;12(4):324-333. doi: 10.2174/1874467212666190719120446. Curr Mol Pharmacol. 2019. PMID: 31677258
-
SIRT6 overexpression induces apoptosis of nasopharyngeal carcinoma by inhibiting NF-κB signaling.Onco Targets Ther. 2018 Nov 1;11:7613-7624. doi: 10.2147/OTT.S179866. eCollection 2018. Onco Targets Ther. 2018. Retraction in: Onco Targets Ther. 2024 Feb 24;17:161-162. doi: 10.2147/OTT.S465407. PMID: 30464510 Free PMC article. Retracted.
-
Identification of RKIP as an invasion suppressor protein in nasopharyngeal carcinoma by proteomic analysis.J Proteome Res. 2008 Dec;7(12):5254-62. doi: 10.1021/pr800602c. J Proteome Res. 2008. PMID: 19367706
Cited by
-
LncRNA NORAD Promotes Vascular Endothelial Cell Injury and Atherosclerosis Through Suppressing VEGF Gene Transcription via Enhancing H3K9 Deacetylation by Recruiting HDAC6.Front Cell Dev Biol. 2021 Jul 9;9:701628. doi: 10.3389/fcell.2021.701628. eCollection 2021. Front Cell Dev Biol. 2021. PMID: 34307380 Free PMC article.
-
HDAC6-mediated deacetylation of FLOT2 maintains stability and tumorigenic function of FLOT2 in nasopharyngeal carcinoma.Zhong Nan Da Xue Xue Bao Yi Xue Ban. 2024 May 28;49(5):687-697. doi: 10.11817/j.issn.1672-7347.2024.240077. Zhong Nan Da Xue Xue Bao Yi Xue Ban. 2024. PMID: 39174882 Free PMC article. Chinese, English.
-
Epigenetic Regulation and Post-Translational Modifications of SNAI1 in Cancer Metastasis.Int J Mol Sci. 2021 Oct 14;22(20):11062. doi: 10.3390/ijms222011062. Int J Mol Sci. 2021. PMID: 34681726 Free PMC article. Review.
References
-
- Lin DC, Meng X, Hazawa M, Nagata Y, Varela AM, Xu L, Sato Y, Liu LZ, Ding LW, Sharma A, Goh BC, Lee SC, Petersson BF, Yu FG, Macary P, Oo MZ, Ha CS, Yang H, Ogawa S, Loh KS, Koeffler HP. The genomic landscape of nasopharyngeal carcinoma. Nat Genet. 2014;46:866–871. - PubMed
-
- Chen YP, Chan ATC, Le QT, Blanchard P, Sun Y, Ma J. Nasopharyngeal carcinoma. Lancet. 2019;394:64–80. - PubMed
-
- Tong ZT, Cai MY, Wang XG, Kong LL, Mai SJ, Liu YH, Zhang HB, Liao YJ, Zheng F, Zhu W, Liu TH, Bian XW, Guan XY, Lin MC, Zeng MS, Zeng YX, Kung HF, Xie D. EZH2 supports nasopharyngeal carcinoma cell aggressiveness by forming a co-repressor complex with HDAC1/HDAC2 and Snail to inhibit E-cadherin. Oncogene. 2012;31:583–594. - PubMed
-
- Salehiniya H MM, Mohammadian-Hafshejani A, Mahdavifar N. Nasopharyngeal cancer in the world: epidemiology, incidence, mortality and risk factors. World Cancer Res J. 2018;5:e1046.
-
- Yu C, Chen L, Yie L, Wei L, Wen T, Liu Y, Chen H. Targeting FoxM1 inhibits proliferation, invasion and migration of nasopharyngeal carcinoma through the epithelialto-mesenchymal transition pathway. Oncol Rep. 2015;33:2402–2410. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
