Observation of arenavirus nucleoprotein heptamer assembly
- PMID: 33534950
- PMCID: PMC8016135
- DOI: 10.1002/2211-5463.13106
Observation of arenavirus nucleoprotein heptamer assembly
Abstract
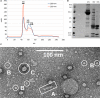
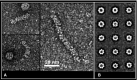
Arenaviruses are enveloped viruses containing a segmented, negative, and ambisense single-stranded RNA genome wrapped with a nucleoprotein (NP). The NP is the most abundant viral protein in infected cells and plays a critical role in both replication/transcription and virion assembly. The NP associates with RNA to form a ribonucleoprotein (RNP) complex, and this implies self-assembly while the exact structure of this polymer is not yet known. Here, we report a measurement of the full-length Mopeia virus NP by negative stain transmission electron microscopy. We observed RNP complex particles with diameter 15 ± 1 nm as well as symmetric circular heptamers of the same diameter, consistent with previous observations.
Keywords: Arenaviridae; Bunyavirales; assembly; nucleoprotein.
© 2021 The Authors. FEBS Open Bio published by John Wiley & Sons Ltd on behalf of Federation of European Biochemical Societies.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures



References
-
- Maes P, Alkhovsky SV, Bào Y, Beer M, Birkhead M, Briese T, Buchmeier MJ, Calisher CH, Charrel RN, Choi IR et al. (2018) Taxonomy of the family Arenaviridae and the order Bunyavirales: update 2018. Arch Virol 163, 2295–2310. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

