Regulation of Fibroblast Cell Polarity by Src Tyrosine Kinase
- PMID: 33535441
- PMCID: PMC7912711
- DOI: 10.3390/biomedicines9020135
Regulation of Fibroblast Cell Polarity by Src Tyrosine Kinase
Abstract
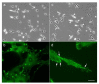
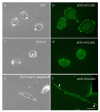
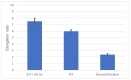
Src protein tyrosine kinases (SFKs) are a family of nonreceptor tyrosine kinases that are localized beneath the plasma membrane and are activated during cell adhesion, migration, and elongation. Due to their involvement in the activation of signal transduction cascades, SFKs have been suggested to play important roles in the determination of cell polarity during cell extension and elongation. However, the mechanism underlying Src-mediated polarity formation remains unclear. The present study was performed to investigate the mechanisms underlying Src-induced cell polarity formation and cell elongation using Src knockout fibroblasts (SYFs) together with an inhibitor of Src. Normal and Src knockout fibroblasts were also transfected with a wild-type c-Src, dominant negative c-Src, or constitutively active c-Src gene to analyze the changes in cell morphology. SYF cells cultured on a glass substrate elongated symmetrically into spindle-shaped cells, with the formation of focal adhesions at both ends of the cells. When normal fibroblasts were treated with Src Inhibitor No. 5, a selective inhibitor of Src tyrosine kinases, they elongated into symmetrical spindle-shaped cells, similar to SYF cells. These results suggest that cell polarity during extension and elongation may be regulated by SFKs and that the expression and regulation of Src are important for the formation of polarity during cell elongation.
Keywords: SFK; c-Src; cell adhesion; cell motility; polarity.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




References
-
- Katoh K., Kano Y., Fujiwara K. Isolation and in vitro contraction of stress fibers. Methods Enzymol. 2000;325:369–380. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

