In-Vitro Antibacterial and Anti-Inflammatory Effects of Surfactin-Loaded Nanoparticles for Periodontitis Treatment
- PMID: 33535497
- PMCID: PMC7912741
- DOI: 10.3390/nano11020356
In-Vitro Antibacterial and Anti-Inflammatory Effects of Surfactin-Loaded Nanoparticles for Periodontitis Treatment
Abstract
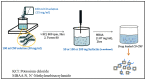
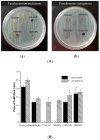
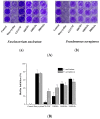
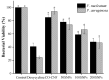
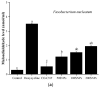
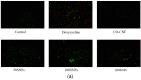
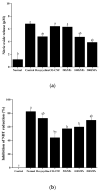
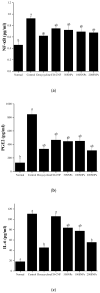
Periodontitis is an inflammatory disease associated with biofilm formation and gingival recession. The practice of nanotechnology in the clinical field is increased overtime due to its potential advantages in drug delivery applications. Nanoparticles can deliver drugs into the targeted area with high efficiency and cause less damages to the tissues. In this study, we investigated the antibacterial and anti-inflammatory properties of surfactin-loaded κ-carrageenan oligosaccharides linked cellulose nanofibers (CO-CNF) nanoparticles. Three types of surfactin-loaded nanoparticles were prepared based on the increasing concentration of surfactin such as 50SNPs (50 mg surfactin-loaded CO-CNF nanoparticles), 100SNPs (100 mg surfactin-loaded CO-CNF nanoparticles), and 200SNPs (200 mg surfactin-loaded CO-CNF nanoparticles). The results showed that the nanoparticles inhibited the growth of Fusobacterium nucleatum and Pseudomonas aeruginosa. The reduction in biofilm formation and metabolic activity of the bacteria were confirmed by crystal violet and MTT assay, respectively. Besides, an increase in oxidative stress was also observed in bacteria. Furthermore, anti-inflammatory effects of surfactin-loaded CO-CNF nanoparticles was observed in lipopolysaccharide (LPS)-stimulated human gingival fibroblast (HGF) cells. A decrease in the production of reactive oxygen species (ROS), transcription factor, and cytokines were observed in the presence of nanoparticles. Collectively, these observations supported the use of surfactin-loaded CO-CNF as a potential candidate for periodontitis management.
Keywords: anti-inflammatory; antibacterial; nanoparticle; periodontitis; surfactin.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




Similar articles
-
Therapeutic effects of antibiotics loaded cellulose nanofiber and κ-carrageenan oligosaccharide composite hydrogels for periodontitis treatment.Sci Rep. 2020 Oct 22;10(1):18037. doi: 10.1038/s41598-020-74845-9. Sci Rep. 2020. PMID: 33093521 Free PMC article.
-
Surfactin-Loaded ĸ-Carrageenan Oligosaccharides Entangled Cellulose Nanofibers as a Versatile Vehicle Against Periodontal Pathogens.Int J Nanomedicine. 2020 Jun 9;15:4021-4047. doi: 10.2147/IJN.S238476. eCollection 2020. Int J Nanomedicine. 2020. PMID: 32606662 Free PMC article.
-
Heme oxygenase-1 signals are involved in preferential inhibition of pro-inflammatory cytokine release by surfactin in cells activated with Porphyromonas gingivalis lipopolysaccharide.Chem Biol Interact. 2010 Dec 5;188(3):437-45. doi: 10.1016/j.cbi.2010.09.007. Epub 2010 Sep 15. Chem Biol Interact. 2010. PMID: 20833156
-
Advanced application of stimuli-responsive drug delivery system for inflammatory arthritis treatment.Mater Today Bio. 2022 Feb 21;14:100223. doi: 10.1016/j.mtbio.2022.100223. eCollection 2022 Mar. Mater Today Bio. 2022. PMID: 35243298 Free PMC article. Review.
-
Surfactin inhibits Fusarium graminearum by accumulating intracellular ROS and inducing apoptosis mechanisms.World J Microbiol Biotechnol. 2023 Oct 12;39(12):340. doi: 10.1007/s11274-023-03790-2. World J Microbiol Biotechnol. 2023. PMID: 37821760 Review.
Cited by
-
Antibacterial Activity of Nanoparticles.Nanomaterials (Basel). 2021 May 25;11(6):1391. doi: 10.3390/nano11061391. Nanomaterials (Basel). 2021. PMID: 34070314 Free PMC article.
-
Algicidal characteristics of novel algicidal compounds, cyclic lipopeptide surfactins from Bacillus tequilensis strain D8, in eliminating Heterosigma akashiwo blooms.Front Microbiol. 2022 Nov 30;13:1066747. doi: 10.3389/fmicb.2022.1066747. eCollection 2022. Front Microbiol. 2022. PMID: 36532506 Free PMC article.
-
Anti-inflammatory and Antioxidant Activity of Neem and Kirata-Induced Silver Nanoparticles Against Oral Biofilm: An In Vitro Study.Cureus. 2024 Aug 25;16(8):e67708. doi: 10.7759/cureus.67708. eCollection 2024 Aug. Cureus. 2024. PMID: 39318902 Free PMC article.
-
Evaluation of Anti-Inflammatory and Antibacterial Properties of Photo-Thermal Hydrogel as Dual Functional Platform for Management of Periodontitis.Int J Nanomedicine. 2025 Mar 11;20:2923-2934. doi: 10.2147/IJN.S508864. eCollection 2025. Int J Nanomedicine. 2025. PMID: 40098724 Free PMC article.
-
Characterization of a λ-Carrageenase Mutant with the Generation of Long-Chain λ-Neocarrageenan Oligosaccharides.Foods. 2024 Jun 18;13(12):1923. doi: 10.3390/foods13121923. Foods. 2024. PMID: 38928863 Free PMC article.
References
-
- Tomofuji T., Irie K., Sanbe T., Azuma T., Ekuni D., Tamaki N., Yamamoto T., Morita M. Periodontitis and increase in circulating oxidative stress. Jpn. Dent. Sci. Rev. 2009;45:46–51. doi: 10.1016/j.jdsr.2008.12.002. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

