ARMC12 regulates spatiotemporal mitochondrial dynamics during spermiogenesis and is required for male fertility
- PMID: 33536340
- PMCID: PMC8017931
- DOI: 10.1073/pnas.2018355118
ARMC12 regulates spatiotemporal mitochondrial dynamics during spermiogenesis and is required for male fertility
Abstract
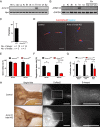
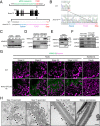
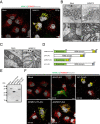
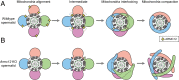
The mammalian sperm midpiece has a unique double-helical structure called the mitochondrial sheath that wraps tightly around the axoneme. Despite the remarkable organization of the mitochondrial sheath, the molecular mechanisms involved in mitochondrial sheath formation are unclear. In the process of screening testis-enriched genes for functions in mice, we identified armadillo repeat-containing 12 (ARMC12) as an essential protein for mitochondrial sheath formation. Here, we engineered Armc12-null mice, FLAG-tagged Armc12 knock-in mice, and TBC1 domain family member 21 (Tbc1d21)-null mice to define the functions of ARMC12 in mitochondrial sheath formation in vivo. We discovered that absence of ARMC12 causes abnormal mitochondrial coiling along the flagellum, resulting in reduced sperm motility and male sterility. During spermiogenesis, sperm mitochondria in Armc12-null mice cannot elongate properly at the mitochondrial interlocking step which disrupts abnormal mitochondrial coiling. ARMC12 is a mitochondrial peripheral membrane protein and functions as an adherence factor between mitochondria in cultured cells. ARMC12 in testicular germ cells interacts with mitochondrial proteins MIC60, VDAC2, and VDAC3 as well as TBC1D21 and GK2, which are required for mitochondrial sheath formation. We also observed that TBC1D21 is essential for the interaction between ARMC12 and VDAC proteins in vivo. These results indicate that ARMC12 uses integral mitochondrial membrane proteins VDAC2 and VDAC3 as scaffolds to link mitochondria and works cooperatively with TBC1D21. Thus, our studies have revealed that ARMC12 regulates spatiotemporal mitochondrial dynamics to form the mitochondrial sheath through cooperative interactions with several proteins on the sperm mitochondrial surface.
Keywords: infertility; mitochondrial sheath formation; sperm mitochondrial dynamics; spermatogenesis.
Copyright © 2021 the Author(s). Published by PNAS.
Conflict of interest statement
The authors declare no competing interest.
Figures



Similar articles
-
Deficiency of the Tbc1d21 gene causes male infertility with morphological abnormalities of the sperm mitochondria and flagellum in mice.PLoS Genet. 2020 Sep 25;16(9):e1009020. doi: 10.1371/journal.pgen.1009020. eCollection 2020 Sep. PLoS Genet. 2020. PMID: 32976492 Free PMC article.
-
Biallelic mutations in ARMC12 cause asthenozoospermia and multiple midpiece defects in humans and mice.J Med Genet. 2023 Feb;60(2):154-162. doi: 10.1136/jmedgenet-2021-108137. Epub 2022 May 9. J Med Genet. 2023. PMID: 35534203
-
TBC1D21 is an essential factor for sperm mitochondrial sheath assembly and male fertility‡.Biol Reprod. 2022 Aug 9;107(2):619-634. doi: 10.1093/biolre/ioac069. Biol Reprod. 2022. PMID: 35403672
-
Haploid male germ cells-the Grand Central Station of protein transport.Hum Reprod Update. 2020 Jun 18;26(4):474-500. doi: 10.1093/humupd/dmaa004. Hum Reprod Update. 2020. PMID: 32318721 Review.
-
Knockout mouse models of sperm flagellum anomalies.Hum Reprod Update. 2006 Jul-Aug;12(4):449-61. doi: 10.1093/humupd/dml013. Epub 2006 Mar 24. Hum Reprod Update. 2006. PMID: 16565154 Review.
Cited by
-
FAM209 associates with DPY19L2, and is required for sperm acrosome biogenesis and fertility in mice.J Cell Sci. 2021 Nov 1;134(21):jcs259206. doi: 10.1242/jcs.259206. Epub 2021 Nov 1. J Cell Sci. 2021. PMID: 34471926 Free PMC article.
-
Disruption of testis-enriched cytochrome c oxidase subunit COX6B2 but not COX8C leads to subfertility.Exp Anim. 2024 Feb 14;73(1):1-10. doi: 10.1538/expanim.23-0055. Epub 2023 Jul 10. Exp Anim. 2024. PMID: 37423748 Free PMC article.
-
CRISPR/Cas9-based genetic screen of SCNT-reprogramming resistant genes identifies critical genes for male germ cell development in mice.Sci Rep. 2021 Jul 29;11(1):15438. doi: 10.1038/s41598-021-94851-9. Sci Rep. 2021. PMID: 34326397 Free PMC article.
-
A novel mechanism of sperm midpiece epididymal maturation and the role of CCDC112 in sperm midpiece formation and establishing an optimal flagella waveform.Cell Commun Signal. 2025 Jul 1;23(1):319. doi: 10.1186/s12964-025-02320-x. Cell Commun Signal. 2025. PMID: 40598224 Free PMC article.
-
Molecular machineries shaping the mitochondrial inner membrane.Nat Rev Mol Cell Biol. 2025 Sep;26(9):706-724. doi: 10.1038/s41580-025-00854-z. Epub 2025 May 14. Nat Rev Mol Cell Biol. 2025. PMID: 40369159 Review.
References
-
- Fawcett D. W., The mammalian spermatozoon. Dev. Biol. 44, 394–436 (1975). - PubMed
-
- Ho H.-C., Wey S., Three dimensional rendering of the mitochondrial sheath morphogenesis during mouse spermiogenesis. Microsc. Res. Tech. 70, 719–723 (2007). - PubMed
-
- Mi Y., Shi Z., Li J., Spata19 is critical for sperm mitochondrial function and male fertility. Mol. Reprod. Dev. 82, 907–913 (2015). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

