Pharmacokinetics and Toxicities of Oral Docetaxel Formulations Co-Administered with Ritonavir in Phase I Trials
- PMID: 33536797
- PMCID: PMC7850405
- DOI: 10.2147/CPAA.S292746
Pharmacokinetics and Toxicities of Oral Docetaxel Formulations Co-Administered with Ritonavir in Phase I Trials
Abstract
Introduction: Docetaxel is widely used as intravenous (IV) chemotherapy. Oral docetaxel is co-administered with the cytochrome P450 3A4 and P-glycoprotein inhibitor ritonavir to increase oral bioavailability. This research explores the relationship between the pharmacokinetics (PK) and toxicity of this novel oral chemotherapy.
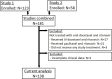
Methods: The patients in two phase I trials were treated with different oral docetaxel formulations in combination with ritonavir in different dose levels, ranging from 20 to 80 mg docetaxel with 100 to 200 mg ritonavir a day. The patients were categorized based on the absence or occurrence of severe treatment-related toxicity (grade ≥3 or any grade leading to treatment alterations). The docetaxel area under the plasma concentration-time curve (AUC) and maximum plasma concentration (Cmax) were associated with toxicity.
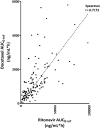
Results: Thirty-four out of 138 patients experienced severe toxicity, most frequently observed as mucositis, fatigue, diarrhea, nausea and vomiting. The severe toxicity group had a significantly higher docetaxel AUC (2231 ± 1405 vs 1011 ± 830 ng/mL*h, p<0.0001) and Cmax (218 ± 178 vs 119 ± 77 ng/mL, p<0.0001) as compared to the patients without severe toxicity. When extrapolated from IV PK data, the patients without severe toxicity had a similar cumulative docetaxel AUC as with standard 3-weekly IV docetaxel, while the Cmax was up to 10-fold lower with oral docetaxel and ritonavir.
Conclusion: Severe toxicity was observed in 25% of the patients treated with oral docetaxel and ritonavir. This toxicity seems related to the PK, as the docetaxel AUC0-inf and Cmax were up to twofold higher in the severe toxicity group as compared to the non-severe toxicity group. Future randomized trials will provide a further evaluation of the toxicity and efficacy of the new weekly oral docetaxel and ritonavir regimen in comparison to standard IV docetaxel.
Keywords: oral docetaxel; pharmacokinetics; ritonavir; toxicity.
© 2021 Vermunt et al.
Conflict of interest statement
Jos Beijnen has received a grant for translational research (ZonMw code 40-41200-98-004) for clinical development of oral taxanes. The two trials that were reviewed in this article were conducted as investigator-initiated phase I trials in the Netherlands Cancer Institute. After their completion, Modra Pharmaceuticals BV was founded as a spin-off company of the Netherlands Cancer Institute, focusing on the further clinical development of oral taxanes co-administrated with ritonavir. Jos H. Beijnen is a part-time employee and shareholder of Modra Pharmaceuticals Holding BV and is a patent holder on oral taxane formulations. The other authors report no conflicts of interest in this work.
Figures
Similar articles
-
ModraDoc006, an oral docetaxel formulation in combination with ritonavir (ModraDoc006/r), in metastatic castration-resistant prostate cancer patients: A phase Ib study.Cancer Rep (Hoboken). 2021 Aug;4(4):e1367. doi: 10.1002/cnr2.1367. Epub 2021 Mar 12. Cancer Rep (Hoboken). 2021. PMID: 33709626 Free PMC article. Clinical Trial.
-
Effect of Food on the Pharmacokinetics of the Oral Docetaxel Tablet Formulation ModraDoc006 Combined with Ritonavir (ModraDoc006/r) in Patients with Advanced Solid Tumours.Drugs R D. 2021 Mar;21(1):103-111. doi: 10.1007/s40268-020-00336-x. Epub 2021 Jan 19. Drugs R D. 2021. PMID: 33464545 Free PMC article. Clinical Trial.
-
Pharmacokinetics of docetaxel and ritonavir after oral administration of ModraDoc006/r in patients with prostate cancer versus patients with other advanced solid tumours.Cancer Chemother Pharmacol. 2021 Jun;87(6):855-869. doi: 10.1007/s00280-021-04259-5. Epub 2021 Mar 20. Cancer Chemother Pharmacol. 2021. PMID: 33744986
-
Lopinavir/ritonavir: a review of its use in the management of HIV infection.Drugs. 2003;63(8):769-802. doi: 10.2165/00003495-200363080-00004. Drugs. 2003. PMID: 12662125 Review.
-
Darunavir: a review of its use in the management of HIV infection in adults.Drugs. 2009;69(4):477-503. doi: 10.2165/00003495-200969040-00007. Drugs. 2009. PMID: 19323590 Review.
Cited by
-
The inhibitory and inducing effects of ritonavir on hepatic and intestinal CYP3A and other drug-handling proteins.Biomed Pharmacother. 2023 Jun;162:114636. doi: 10.1016/j.biopha.2023.114636. Epub 2023 Apr 1. Biomed Pharmacother. 2023. PMID: 37004323 Free PMC article. Review.
-
Ritonavir-Boosted Exposure of Kinase Inhibitors: an Open Label, Cross-over Pharmacokinetic Proof-of-Concept Trial with Erlotinib.Pharm Res. 2022 Apr;39(4):669-676. doi: 10.1007/s11095-022-03244-8. Epub 2022 Mar 29. Pharm Res. 2022. PMID: 35352280 Free PMC article.
-
Impact of loperamide on the pharmacokinetics and tissue disposition of ritonavir-boosted oral docetaxel therapy; a preclinical assessment.Cancer Chemother Pharmacol. 2024 Jul;94(1):79-87. doi: 10.1007/s00280-024-04662-8. Epub 2024 Mar 8. Cancer Chemother Pharmacol. 2024. PMID: 38456955
-
Pharmacological Potential of Lathyrane-Type Diterpenoids from Phytochemical Sources.Pharmaceuticals (Basel). 2022 Jun 23;15(7):780. doi: 10.3390/ph15070780. Pharmaceuticals (Basel). 2022. PMID: 35890079 Free PMC article. Review.
References
LinkOut - more resources
Full Text Sources
Other Literature Sources