Computational Derivation of Core, Dynamic Human Blunt Trauma Inflammatory Endotypes
- PMID: 33537029
- PMCID: PMC7848165
- DOI: 10.3389/fimmu.2020.589304
Computational Derivation of Core, Dynamic Human Blunt Trauma Inflammatory Endotypes
Abstract
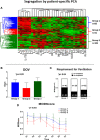
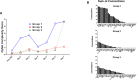
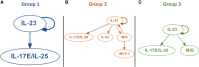
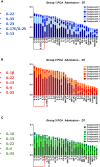
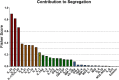
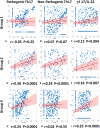
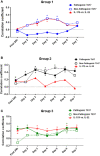
Systemic inflammation ensues following traumatic injury, driving immune dysregulation and multiple organ dysfunction (MOD). While a balanced immune/inflammatory response is ideal for promoting tissue regeneration, most trauma patients exhibit variable and either overly exuberant or overly damped responses that likely drive adverse clinical outcomes. We hypothesized that these inflammatory phenotypes occur in the context of severe injury, and therefore sought to define clinically distinct endotypes of trauma patients based on their systemic inflammatory responses. Using Patient-Specific Principal Component Analysis followed by unsupervised hierarchical clustering of circulating inflammatory mediators obtained in the first 24 h after injury, we segregated a cohort of 227 blunt trauma survivors into three core endotypes exhibiting significant differences in requirement for mechanical ventilation, duration of ventilation, and MOD over 7 days. Nine non-survivors co-segregated with survivors. Dynamic network inference, Fisher Score analysis, and correlations of IL-17A with GM-CSF, IL-10, and IL-22 in the three survivor sub-groups suggested a role for type 3 immunity, in part regulated by Th17 and γδ 17 cells, and related tissue-protective cytokines as a key feature of systemic inflammation following injury. These endotypes may represent archetypal adaptive, over-exuberant, and overly damped inflammatory responses.
Keywords: biomarker; critical illness; inflammation; network analysis; systems biology.
Copyright © 2021 Schimunek, Lindberg, Cohen, Namas, Mi, Yin, Barclay, El-Dehaibi, Abboud, Zamora, Billiar and Vodovotz.
Conflict of interest statement
YV is a co-founder of, and stakeholder in, Immunetrics, Inc. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Similar articles
-
Individual-specific principal component analysis of circulating inflammatory mediators predicts early organ dysfunction in trauma patients.J Crit Care. 2016 Dec;36:146-153. doi: 10.1016/j.jcrc.2016.07.002. Epub 2016 Jul 11. J Crit Care. 2016. PMID: 27546764 Free PMC article.
-
Diurnal Variation in Systemic Acute Inflammation and Clinical Outcomes Following Severe Blunt Trauma.Front Immunol. 2019 Nov 20;10:2699. doi: 10.3389/fimmu.2019.02699. eCollection 2019. Front Immunol. 2019. PMID: 31824494 Free PMC article.
-
Computational evidence for an early, amplified systemic inflammation program in polytrauma patients with severe extremity injuries.PLoS One. 2019 Jun 4;14(6):e0217577. doi: 10.1371/journal.pone.0217577. eCollection 2019. PLoS One. 2019. PMID: 31163056 Free PMC article.
-
The Use of Multiplexing to Identify Cytokine and Chemokine Networks in the Immune-Inflammatory Response to Trauma.Antioxid Redox Signal. 2021 Dec;35(16):1393-1406. doi: 10.1089/ars.2021.0054. Epub 2021 May 19. Antioxid Redox Signal. 2021. PMID: 33860683 Free PMC article. Review.
-
Complement After Trauma: Suturing Innate and Adaptive Immunity.Front Immunol. 2018 Sep 24;9:2050. doi: 10.3389/fimmu.2018.02050. eCollection 2018. Front Immunol. 2018. PMID: 30319602 Free PMC article. Review.
Cited by
-
A common single nucleotide polymorphism is associated with inflammation and critical illness outcomes.iScience. 2023 Oct 28;26(12):108333. doi: 10.1016/j.isci.2023.108333. eCollection 2023 Dec 15. iScience. 2023. PMID: 38034362 Free PMC article.
-
Interleukin-17 as a spatiotemporal bridge from acute to chronic inflammation: Novel insights from computational modeling.WIREs Mech Dis. 2023 May-Jun;15(3):e1599. doi: 10.1002/wsbm.1599. Epub 2023 Jan 29. WIREs Mech Dis. 2023. PMID: 36710253 Free PMC article.
-
Towards systems immunology of critical illness at scale: from single cell 'omics to digital twins.Trends Immunol. 2023 May;44(5):345-355. doi: 10.1016/j.it.2023.03.004. Epub 2023 Mar 24. Trends Immunol. 2023. PMID: 36967340 Free PMC article. Review.
-
What's next for computational systems biology?Front Syst Biol. 2023 Sep 19;3:1250228. doi: 10.3389/fsysb.2023.1250228. eCollection 2023. Front Syst Biol. 2023. PMID: 40809471 Free PMC article. Review.
-
Plasma proteomics reveals early, broad release of chemokine, cytokine, TNF, and interferon mediators following trauma with delayed increases in a subset of chemokines and cytokines in patients that remain critically ill.Front Immunol. 2022 Nov 30;13:1038086. doi: 10.3389/fimmu.2022.1038086. eCollection 2022. Front Immunol. 2022. PMID: 36532045 Free PMC article.
References
-
- Namas R, Mi Q, Namas R, Almahmoud K, Zaaqoq A, Abdul Malak O, et al. Insights into the role of chemokines, damage-associated molecular patterns, and lymphocyte-derived mediators from computational models of trauma-induced inflammation. Antioxid Redox Signaling (2015) 10:1370–87. 10.1089/ars.2015.6398 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

