Highly specific and label-free histological identification of microcrystals in fresh human gout tissues with stimulated Raman scattering
- PMID: 33537075
- PMCID: PMC7847673
- DOI: 10.7150/thno.53755
Highly specific and label-free histological identification of microcrystals in fresh human gout tissues with stimulated Raman scattering
Abstract
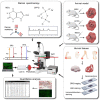
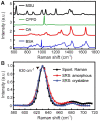
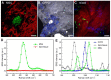
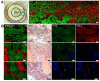
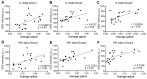
Gout is a common metabolic disease with growing burden, caused by monosodium urate (MSU) microcrystal deposition. In situ and chemical-specific histological identification of MSU is crucial in the diagnosis and management of gout, yet it remains inaccessible for current histological methods. Methods: Stimulated Raman scattering (SRS) microscopy was utilized to image MSU based on its fingerprint Raman spectra. We first tested SRS for the diagnosis capability of gout and the differentiation power from pseudogout with rat models of acute gout arthritis, calcium pyrophosphate deposition disease (CPDD) and comorbidity. Then, human synovial fluid and surgical specimens (n=120) were were imaged with SRS to obtain the histopathology of MSU and collagen fibers. Finally, quantitative SRS analysis was performed in gout tissue of different physiological phases (n=120) to correlate with traditional histopathology including H&E and immunohistochemistry staining. Results: We demonstrated that SRS is capable of early diagnosis of gout, rapid detection of MSU in synovial fluid and fresh unprocessed surgical tissues, and accurate differentiation of gout from pseudogout in various pathophysiological conditions. Furthermore, quantitative SRS analysis revealed the optical characteristics of MSU deposition at different pathophysiological stages, which were found to matched well with corresponding immunofluorescence histochemistry features. Conclusion: Our work demonstrated the potential of SRS microscopy for rapid intraoperative diagnosis of gout and may facilitate future fundamental researches of MSU-based diseases.
Keywords: gout; label-free histology; monosodium urate; stimulated Raman scattering.
© The author(s).
Conflict of interest statement
Competing Interests: The authors have declared that no competing interest exists.
Figures





References
-
- Kuo CF, Grainge MJ, Zhang W, Doherty M. Global epidemiology of gout: prevalence, incidence and risk factors. Nat Rev Rheumatol. 2015;11:649–62. - PubMed
-
- Zhu Y, Pandya BJ, Choi HK. Prevalence of gout and hyperuricemia in the US general population: The national health and nutrition examination survey 2007-2008. Arthritis Rheum. 2011;63:3136–41. - PubMed
-
- Dalbeth N, Merriman TR, Stamp LK. Gout. Lancet. 2016;388:2039–52. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

