Fundus Autofluorescence in Neovascular Age-Related Macular Degeneration: A Clinicopathologic Correlation Relevant to Macular Atrophy
- PMID: 33540168
- PMCID: PMC9631811
- DOI: 10.1016/j.oret.2021.01.012
Fundus Autofluorescence in Neovascular Age-Related Macular Degeneration: A Clinicopathologic Correlation Relevant to Macular Atrophy
Abstract
Purpose: Macular atrophy (MA) of retinal pigment epithelium (RPE) and photoreceptors leads to vision loss in neovascular age-related macular degeneration (nAMD) despite successful treatment with antiangiogenic agents. To enhance understanding of MA, fortify the cellular basis of fundus autofluorescence (FAF) imaging, and inform management of nAMD, we performed histologic analysis of an eye with multimodal clinical imaging and apparent prior exudation due to nAMD.
Design: Case study and clinicopathologic correlation.
Participant: A White woman in whom age-related macular degeneration (AMD) findings of inactive subretinal fibrosis (right eye) were followed for 9 years using FAF and OCT, with no detectable subretinal fluid or other recurrent exudation and no intravitreal injections before her death at age 90 years.
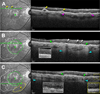
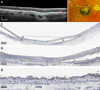
Methods: The right eye was preserved 6.25 hours after death, postfixed in osmium tannic acid paraphenylenediamine, and prepared for submicrometer epoxy resin sections (n = 115), with 19 matched to clinical OCT B-scans.
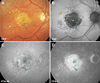
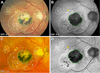
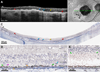
Main outcome measures: Light microscopic morphology of a hyperautofluorescent (hyperFAF) area due to prior exudation ("floodplain" hyperFAF), hypoautofluorescent (hypoFAF) spots of MA, and areas of unremarkable FAF.
Results: Floodplain hyperFAF was visible throughout the 9 years of follow-up, with several hypoFAF atrophic spots expanding within it over time. The hyperFAF pattern corresponded to outer retinal atrophy (ORA) on OCT and photoreceptor loss over dysmorphic yet continuous RPE in histology. The hypoFAF spots inside the floodplain corresponded to complete RPE and outer retinal atrophy (cRORA) on OCT and loss of both photoreceptors and RPE in histology. In contrast, areas of unremarkable FAF showed continuous RPE accompanied by full-length photoreceptors and a thick outer nuclear layer.
Conclusions: This direct clinicopathologic correlation for FAF imaging is the first for nAMD. Fundus autofluorescence is a projection image that involves optical signal modulation by photoreceptors as well as emission signal sources in RPE. Hyperautofluorescence due to an exudative floodplain signifies loss of photoreceptors over continuous RPE. Hypoautofluorescence in MA signifies loss of both cell layers. For maximal value, fundus autofluorescence imaging should be interpreted with the multilayer perspective provided by OCT. Prevention of exudation in nAMD may preserve photoreceptors.
Keywords: Age-related macular degeneration; Autofluorescence; Histopathology; Lipofuscin; Neovascularization; Photoreceptors; Retinal pigment epithelium.
Copyright © 2021 American Academy of Ophthalmology. Published by Elsevier Inc. All rights reserved.
Figures

References
-
- Bhisitkul RB, Mendes TS, Rofagha S, et al. Macular atrophy progression and 7-year vision outcomes in subjects from the ANCHOR, MARINA, and HORIZON studies: the SEVEN-UP study. Am J Ophthalmol 2015;159(5):915–24 e2. - PubMed
-
- Sadda SR, Tuomi LL, Ding B, et al. Macular Atrophy in the HARBOR Study for Neovascular Age-Related Macular Degeneration. Ophthalmology 2018;125(6):878–86. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

