Flexibility and Adaptation of Cancer Cells in a Heterogenous Metabolic Microenvironment
- PMID: 33540663
- PMCID: PMC7867260
- DOI: 10.3390/ijms22031476
Flexibility and Adaptation of Cancer Cells in a Heterogenous Metabolic Microenvironment
Abstract
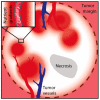
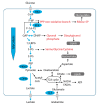
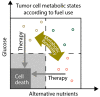
The metabolic microenvironment, comprising all soluble and insoluble nutrients and co-factors in the extracellular milieu, has a major impact on cancer cell proliferation and survival. A large body of evidence from recent studies suggests that tumor cells show a high degree of metabolic flexibility and adapt to variations in nutrient availability. Insufficient vascular networks and an imbalance of supply and demand shape the metabolic tumor microenvironment, which typically contains a lower concentration of glucose compared to normal tissues. The present review sheds light on the recent literature on adaptive responses in cancer cells to nutrient deprivation. It focuses on the utilization of alternative nutrients in anabolic metabolic pathways in cancer cells, including soluble metabolites and macromolecules and outlines the role of central metabolic enzymes conferring metabolic flexibility, like gluconeogenesis enzymes. Moreover, a conceptual framework for potential therapies targeting metabolically flexible cancer cells is presented.
Keywords: adaptability; cancer; gluconeogenesis; heterogeneity; metabolic microenvironment.
Conflict of interest statement
The authors declare no conflict of interests.
Figures

References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

